Serum VEGF level is associated with the outcome of patients with hepatocellular carcinoma: a meta-analysis
Introduction
Hepatocellular carcinoma (HCC) is the sixth most common cancer and the third most common cause of death from cancer and the third largest cause of cancer-related deaths worldwide (1). Although liver resection is an effective treatment for patients with advanced HCC, the long-term postoperative prognosis remains poor because of the high recurrence rate and lack of effective systemic therapy for HCC patients with metastases. The main prognostic factors are clinicopathological characteristics of the disease, including tumor size, stage, and grade. However, the prognostic factors do not fully predict individual clinical outcome. There is the need for better markers to identify patients with poor prognosis at the time of diagnosis. Researches have focused on the potential role of new biological factors involved in the carcinogenic process as prognostic markers in patients with HCC.
Angiogenesis, the formation of new blood vessels from existing vasculature, is an important process in many malignancies including HCC. It is the result of an intricate balance between pro-angiogenic and anti-angiogenic factors. VEGF (also referred to as VEGF-A, vascular permeability factor) is a critical pro-angiogenic factor in cancer. The role of VEGF in the regulation of angiogenesis is the object of intense investigation for more than a decade. The VEGF family is composed of several subtypes, including VEGF-A, VEGF-B, VEGF-C and VEGF-D which exist as numerous splice variant isoforms (2,3). Many anti-angiogenic compounds are being developed, most of which target VEGF and/or its receptors. It is necessary to establish whether VEGF expression is a prognostic marker in HCC.
Many studies have evaluated whether serum VEGF level may be a prognostic factor for survival in patients with HCC. However, the results of the studies are inconclusive and no consensus has been reached. It is unknown whether differences in these investigations have been mostly due to their limited sample size or genuine heterogeneity. Thus, we conducted a meta-analysis of all available studies relating serum VEGF level with the clinical outcome in patients with HCC.
Materials and methods
Search strategy and study selection
The electronic databases PubMed and CNKI (China National Knowledge Infrastructure) were searched for studies to include in the present meta-analysis. An upper date limit of June 20, 2013 was applied; we used no lower date limit. Searches included the terms “hepatocellular or liver”, “cancer or carcinoma or tumor or neoplasm”, “VEGF”, “vascular endothelial growth factor”, and “prognosis”. We also reviewed the Cochrane Library for relevant articles. The references reported in the identified studies were also used to complete the search.
Studies eligible for inclusion in this meta-analysis met the following criteria: (I) measure VEGF expression in the primary hepatocellular carcinoma serum with RT-PCR or ELISA (reverse transcription-polymerase chain reaction/enzyme linked immunosorbent assay); (II) provide information on survival [i.e., disease free survival (DFS) and/or overall survival (OS), studies investigating response rates only were excluded] and (III) When the same author reported results obtained from the same patient population in more than one publication, only the most recent report, or the most complete one, was included in the analysis. Two reviewers (P.Z. and Q.Q.) independently determined study eligibility. Disagreements were resolved by consensus.
Data extraction and quality assessment
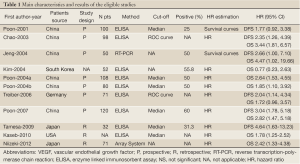
Data retrieved from the reports included author, publication year, patient source, study design, test method, definition of positivity (cut-off value), VEGF positive ratio and survival data (Table 1). If data from any of the above categories were not reported in the primary study, items were treated as “not applicable”. We did no contact the author of the primary study to request the information. We did not use prespecified quality-related inclusion or exclusion criteria and did not weigh each study by a quality score, because the quality score has not received general agreement for use in a meta-analysis, especially observational studies (4). The data extraction and quality assessment could refer to our previous published meta-analysis (5-8).
Full Table
Statistical methods
Included studies were divided into two groups for analysis: those with data regarding OS and those regarding DFS. For the quantitative aggregation of the survival results, we measured the impact of serum VEGF level on survival by HR between the two survival distributions. HRs and 95% confidence intervals (CIs) were used to combine as the effective value. If the HRs and their 95% CIs were given explicitly in the articles, we used crude ones. When these variables were not given explicitly, they were calculated from the available numerical data using methods reported by Parmar et al. (9).
Heterogeneity of the individual HRs was calculated with χ2 tests according to Peto’s method (10). Heterogeneity test with inconsistency index (Ι2) statistic and Q statistic was performed. If HRs were found to have fine homogeneity, a fixed effect model was used for secondary analysis; if not, a random-effect model was used. DerSimonian-Laird random effects analysis (11) was used to estimate the effect of serum VEGF high level on survival. By convention, an observed HR>1 implies worse survival for the group with serum VEGF high level. The impact of VEGF on survival was considered to be statistically significant if the 95% confidence interval (CI) did not overlap with 1. Horizontal lines represent 95% CIs. Each box represents the HR point estimate, and its area is proportional to the weight of the study. The diamond (and broken line) represents the overall summary estimate, with CI represented by its width. The unbroken vertical line is set at the null value (HR=1.0).
Evidence of publication bias was sought using the methods of Egger et al. (12) and of Begg et al. (13). Intercept significance was determined by the t test suggested by Egger (P<0.05 was considered representative of statistically significant publication bias). All of the calculations were performed by STATA version 11.0 (Stata Corporation, College Station, TX).
Results
Study selection and characteristics
Fourteen studies (14-24) published between 2001 and 2012 were eligible for this meta-analysis. All reported the prognostic value of serum VEGF level for survival in hepatocellular carcinoma patients. The total number of patients included was 782, ranging from 32 to 120 patients per study (median 78). The major characteristics of the 11 eligible publications are reported in Table 1. The studies were conducted in 5 countries (China, Japan, South Korea, Germany and USA).
All of the studies reported the prognostic value of serum VEGF level for survival in patients with HCC tissue. Of the 11 studies, 9 directly reported HRs (multivariate analysis), while the other 2 studies provided survival curves. Among them, the proportion of patients exhibiting serum high VEGF level in individual studies ranged from 25% to 60%. Estimation using survival curves were segregated according to either OS or DFS. A HR on DFS and OS could be extracted for 6 publications and 9 publications of studies, respectively. Eight of the 11 studies identified high VEGF level as an indicator of poor prognosis, and the other 3 studies showed no statistically significant impact of high VEGF level on survival.
Meta-analysis
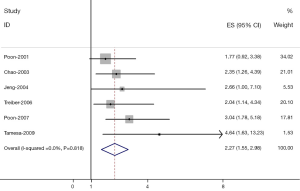
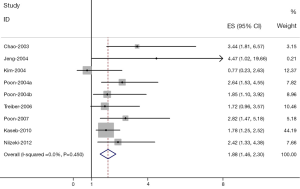
The results of the meta-analysis were shown in Table 2 and Figures 1,2. Overall, the combined HR for all 6 eligible studies evaluating high VEGF level on DFS was 2.27 (95% CI: 1.55-2.98), suggesting that high VEGF level in serum was an indicator of poor prognosis for hepatocellular carcinoma. No significant heterogeneity was observed among the studies (Q=7.39, I2=0%, P=0.818). In addition, for OS analysis, statistically significant effect of high VEGF level (HR=1.88, 95% CI: 1.46-2.30) in patients with HCC was also observed. (Q=2.48, I2=0.0%, P=0.450).

Full Table
Publication bias
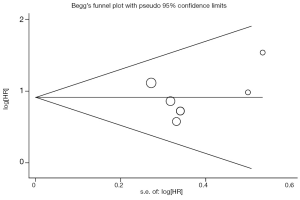
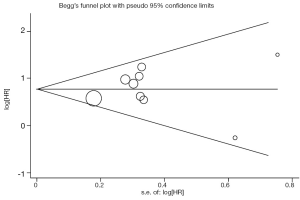
Begg’s funnel plot and Egger’s test were performed to assess the publication bias in the literature. All 6 eligible studies investigating high VEGF level on DFS yielded a Begg’s test score of P=0.707 and an Egger’s test score of P=0.537, meanwhile according to the funnel plot (Figure 3), the absence of publication bias was found. Moreover, the absence of publication biases were found for investigating high VEGF level on OS (a Begg’s test score of P=0.917 and an Egger’s test score of P=0.699) (Figure 4).
Discussion
Members of the VEGF family promote two very important processes in vivo, angiogenesis and lymphangiogenesis, which involve growth of new blood and lymphatic vessels from pre-existing vasculature, respectively. VEGF-A exists as a homodimer or can heterodimerize with either VEGF-B or non-VEGF factors such as placenta growth factor (PIGF) (25-27). VEGF-A and VEGF-B promote vascular angiogenesis primarily through activation of vascular endothelial cell associated VEGFR-1 (Flt1) and VEGFR-2 (Flk1/KDR). On the other hand, VEGF-C and VEGF-D which are ligands for VEGFR-2 and VEGFR-3, promote angiogenesis and lymphangiogenesis (28,29).
The present meta-analysis has combined 11 publications including 782 patients to yield statistics, indicating a statistically significant role of VEGF on overall survival, and disease-free survival in HCC. Our data were consistent with the results of a previous meta-analysis (30) published in 2009 that showed an association between VEGF overexpression and poor survival of patients with HCC. This analysis (30) included only 8 studies. We have improved upon that previous meta-analysis by including more recent related studies and by generally using a more comprehensive search strategy. Screening, study selection and quality assessment were performed independently and reproducibly by two reviewers. We also explored heterogeneity and potential publication bias in accordance with published guidelines.
There were several meta-analyses studying the prognostic value of VEGF in other cancer types, such as head and neck squamous cancer (31), lung cancer (32), colon cancer (33), gastric cancer (34), and hepatocellular carcinoma (35). Association of VEGF overexpression with poor outcomes provides a rationale for anti-angiogenics use in the treatment of cancer. VEGF has become a leading therapeutic target for the treatment of cancer. Potentially therapeutic strategies to inhibit VEGF pathway include monoclonal antibodies directed against VEGF, tyrosine kinase inhibitors (TKIs), and antisense strategies (36). Bevacizumab (Avastin) is a humanized monoclonal antibody directed against VEGF (37). It binds to all isoforms of VEGF-A, thus blocking its binding to VEGFR, but it does not bind to other VEGF molecules, such as VEGF-B or VEGF-C.
The heterogeneity issue was complicated in the systematic review and meta-analysis was. We found no significant heterogeneity among all studies included and subgroup analysis. Another potential source of bias is related to the method of HR and 95% CI extrapolation. If these statistics were not reported by the authors, we calculated them from the data available in the article. If this was not possible, we extrapolated them from the survival curves, necessarily making assumptions about the censoring process. Data for multivariate survival analysis reported in the article were included in the present systematic review and meta-analysis; if these data were not available, data calculated from survival curves by univariate analysis were included. These results should be confirmed by an adequately designed prospective study. Furthermore, the exact value of VEGF overexpression status needs to be determined by appropriate multivariate analysis.
Publication bias (38) is a major concern for all forms of meta-analysis; positive results tend to be accepted by journals, while negative results are often rejected or not even submitted. The present analysis does not support publication bias; the obtained summary statistics likely approximate the actual average. However, it should be noted that our meta-analysis could not completely exclude biases. For example, the study was restricted to papers published in English and Chinese, which probably introduced bias.
In conclusion, our meta-analysis estimated the association between prognostic significance of serum high VEGF level and patients with HCC. As determined in our meta-analysis, we concluded that serum high VEGF level was associated with poor overall survival and disease-free survival, and there is no significant heterogeneity among all studies. To strengthen our findings, well-designed prospective studies with better standardized assessment of prognostic markers should help to explore the relation between serum VEGF level and the outcome of patients with HCC.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Greten TF, Manns MP, Korangy F. Immunotherapy of HCC. Rev Recent Clin Trials 2008;3:31-9. [PubMed]
- Ferrara N. VEGF and the quest for tumour angiogenesis factors. Nat Rev Cancer 2002;2:795-803. [PubMed]
- Hicklin DJ, Ellis LM. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol 2005;23:1011-27. [PubMed]
- Altman DG. Systematic reviews of evaluations of prognostic variables. BMJ 2001;323:224-8. [PubMed]
- Zhan P, Qian Q, Yu LK. Prognostic value of COX-2 expression in patients with non-small cell lung cancer: a systematic review and meta-analysis. J Thorac Dis 2013;5:40-7. [PubMed]
- Zhan P, Qian Q, Wan B, et al. Prognostic value of TTF-1 expression in patients with non-small cell lung cancer: a meta-analysis. Transl Cancer Res 2013;2:25-32.
- Zhan P, Wang Q, Qian Q, et al. Megestrol acetate in cancer patients with anorexia-cachexia syndrome: a meta-analysis. Transl Cancer Res 2013;2:74-9.
- Zhan P, Wang Q, Qian Q, et al. Risk of venous thromboembolism with the erythropoiesis-stimulating agents (ESAs) for the treatment of cancer-associated anemia: a meta-analysis of randomized control trials. Chin Clin Oncol 2012;1:19.
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med 1998;17:2815-34. [PubMed]
- Yusuf S, Peto R, Lewis J, et al. Beta blockade during and after myocardial infarction: an overview of the randomized trials. Prog Cardiovasc Dis 1985;27:335-71. [PubMed]
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7:177-88. [PubMed]
- Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315:629-34. [PubMed]
- Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994;50:1088-101. [PubMed]
- Chao Y, Li CP, Chau GY, et al. Prognostic significance of vascular endothelial growth factor, basic fibroblast growth factor, and angiogenin in patients with resectable hepatocellular carcinoma after surgery. Ann Surg Oncol 2003;10:355-62. [PubMed]
- Jeng KS, Sheen IS, Wang YC, et al. Prognostic significance of preoperative circulating vascular endothelial growth factor messenger RNA expression in resectable hepatocellular carcinoma: a prospective study. World J Gastroenterol 2004;10:643-8. [PubMed]
- Poon RT, Lau C, Pang R, et al. High serum vascular endothelial growth factor levels predict poor prognosis after radiofrequency ablation of hepatocellular carcinoma: importance of tumor biomarker in ablative therapies. Ann Surg Oncol 2007;14:1835-45. [PubMed]
- Poon RT, Ng IO, Lau C, et al. Serum vascular endothelial growth factor predicts venous invasion in hepatocellular carcinoma: a prospective study. Ann Surg 2001;233:227-35. [PubMed]
- Treiber G, Wex T, Röcken C, et al. Impact of biomarkers on disease survival and progression in patients treated with octreotide for advanced hepatocellular carcinoma. J Cancer Res Clin Oncol 2006;132:699-708. [PubMed]
- Kim SJ, Choi IK, Park KH, et al. Serum vascular endothelial growth factor per platelet count in hepatocellular carcinoma: correlations with clinical parameters and survival. Jpn J Clin Oncol 2004;34:184-90. [PubMed]
- Poon RT, Ho JW, Tong CS, et al. Prognostic significance of serum vascular endothelial growth factor and endostatin in patients with hepatocellular carcinoma. Br J Surg 2004;91:1354-60. [PubMed]
- Poon RT, Lau C, Yu WC, et al. High serum levels of vascular endothelial growth factor predict poor response to transarterial chemoembolization in hepatocellular carcinoma: a prospective study. Oncol Rep 2004;11:1077-84. [PubMed]
- Tamesa T, Iizuka N, Mori N, et al. High serum levels of vascular endothelial growth factor after hepatectomy are associated with poor prognosis in hepatocellular carcinoma. Hepatogastroenterology 2009;56:1122-6. [PubMed]
- Kaseb AO, Hassan MM, Lin E, et al. V-CLIP: Integrating plasma vascular endothelial growth factor into a new scoring system to stratify patients with advanced hepatocellular carcinoma for clinical trials. Cancer 2010. [Epub ahead of print]. [PubMed]
- Niizeki T, Sumie S, Torimura T, et al. Serum vascular endothelial growth factor as a predictor of response and survival in patients with advanced hepatocellular carcinoma undergoing hepatic arterial infusion chemotherapy. J Gastroenterol 2012;47:686-95. [PubMed]
- Parikh AA, Ellis LM. The vascular endothelial growth factor family and its receptors. Hematol Oncol Clin North Am 2004;18:951-71. [PubMed]
- Xie K, Wei D, Shi Q, et al. Constitutive and inducible expression and regulation of vascular endothelial growth factor. Cytokine Growth Factor Rev 2004;15:297-324. [PubMed]
- Hicklin DJ, Ellis LM. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol 2005;23:1011-27. [PubMed]
- Lee J, Gray A, Yuan J, et al. Vascular endothelial growth factor-related protein: a ligand and specific activator of the tyrosine kinase receptor Flt4. Proc Natl Acad Sci U S A 1996;93:1988-92. [PubMed]
- Jeltsch M, Kaipainen A, Joukov V, et al. Hyperplasia of lymphatic vessels in VEGF-C transgenic mice. Science 1997;276:1423-5. [PubMed]
- Schoenleber SJ, Kurtz DM, Talwalkar JA, et al. Prognostic role of vascular endothelial growth factor in hepatocellular carcinoma: systematic review and meta-analysis. Br J Cancer 2009;100:1385-92. [PubMed]
- Kyzas PA, Cunha IW, Ioannidis JP. Prognostic significance of vascular endothelial growth factor immunohistochemical expression in head and neck squamous cell carcinoma: a meta-analysis. Clin Cancer Res 2005;11:1434-40. [PubMed]
- Zhan P, Wang J, Lv XJ, et al. Prognostic value of vascular endothelial growth factor expression in patients with lung cancer: a systematic review with meta-analysis. J Thorac Oncol 2009;4:1094-103. [PubMed]
- Des Guetz G, Uzzan B, Nicolas P, et al. Microvessel density and VEGF expression are prognostic factors in colorectal cancer. Meta-analysis of the literature. Br J Cancer 2006;94:1823-32. [PubMed]
- Peng L, Zhan P, Zhou Y, et al. Prognostic significance of vascular endothelial growth factor immunohistochemical expression in gastric cancer: a meta-analysis. Mol Biol Rep 2012;39:9473-84. [PubMed]
- Schoenleber SJ, Kurtz DM, Talwalkar JA, et al. Prognostic role of vascular endothelial growth factor in hepatocellular carcinoma: systematic review and meta-analysis. Br J Cancer 2009;100:1385-92. [PubMed]
- Kowanetz M, Ferrara N. Vascular endothelial growth factor signaling pathways: therapeutic perspective. Clin Cancer Res 2006;12:5018-22. [PubMed]
- Ferrara N, Hillan KJ, Gerber HP, et al. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat Rev Drug Discov 2004;3:391-400. [PubMed]
- Begg CB, Berlin JA. Publication bias: A problem in interpreting medical data. J R Stat Soc A 1988;151:419-63.


