Survival after resection of colorectal liver metastases in octogenarians and sexagenarians compared to their respective age-matched national population
Introduction
In 15 years, the worldwide population of 80- to 89-year-old individuals (octogenarians) has increased by almost 80% (census.gov U.S. Census Bureau, International Data Base). The incidence of colorectal cancer increases with age and many patients present with liver metastases (1,2). As a consequence, more octogenarians are likely to be referred for resection of colorectal liver metastases (CLM).
Current literature describing survival after resection of CLM in elderly patients, especially in octogenarians, is sparse (3-7). Existing data may be limited by several factors. The majority of studies define elderly as 70 years or more. This may be inaccurate, as an increasing number of patients are as physically and mentally fit as younger patients (8-13). Furthermore, at the time of diagnosis of colorectal cancer, the median age is close to 70 years. Defining nearly half of the diseased population as elderly may not be statistically ideal (1). Most important, the impact of age on survival has not been investigated in patients resected for CLM and to date; no study has compared the survival of resected patients to the expected survival of the age-matched national population.
The aim of the current study was to investigate survival in octogenarians undergoing resection of CLM. Sexagenarian patients (60 to 69 years of age and representing the institutional average-age patient undergoing resection of CLM) were selected as controls. Survival curves were compared between patients and their age-matched national background population to depict the deviation and thereby the impact of age.
Methods
Study population
The current study was approved by the internal review board (protocol number 2015/18666). The manuscript was prepared according to the STROBE guidelines (http://www.strobe-statement.org). Patients who underwent liver resection in the time period from 2002 through 2014 were identified from the institutional liver database. Octogenarians were defined as patients between the age of 80 and 89 and were included in retrospective analyses. The following data were extracted and analyzed: age, primary tumor lymph node status, number of liver metastases, diameter of the largest liver metastases, the level of carcinoembryonic antigen (CEA), the American Society of Anesthesiologists (ASA) Class, comorbidities (heart, lung, diabetes, or other significant comorbidity), type of liver resection (major, three segments or more; minor, two segments or less), positive resection margin status (R1, defined as less than 1 mm distance from tumor tissue to resection margin), postoperative interventions for complications (drain placement, blood transfusion, reoperation, or other significant postoperative complication), the use of preoperative chemotherapy, recurrence-free survival (RFS), and overall survival (OS). RFS was defined as days from liver resection to the diagnosis of recurrence (hepatic or extrahepatic) and OS defined as days from liver resection to death.
Patient management
Triphasic computed tomography (CT) of the chest and abdomen, and magnetic resonance imaging (MRI) of the liver were used to assess resectability and extrahepatic disease. The following regiments of perioperative chemotherapy were administered in selected patients: Nordic Fluorouracil Leucovorin (FLV), Nordic Fluorouracil Leucovorin Oxaliplatin (FLOX), Nordic Fluorouracil Leucovorin Irinotecan (FLIRI). The number of octogenarians receiving chemotherapy and the heterogeneity regarding the regiments precluded further analyses. Liver resection was not performed in the presence of unresectable extrahepatic disease. All patients underwent preoperative outpatient examination by a hepatobiliary surgeon and anesthesiologist to determine if the patient was fit for liver resection. In general, the patient had to be self-reliant with respect to nutrition and mobilization, ability to live independently or alone without the need for home nursing, be mentally healthy, and without major cardiopulmonary disease. However, the preoperative screening was not performed systematically nor recorded prospectively and the final decision to accept for surgery was at the discretion of the surgeon.
Both laparoscopic and open liver resection were performed and intraoperative ultrasonography was used in selected patients to assess previously known and undetected lesions. Parenchymal sparing liver resection was the method of choice and performed whenever possible. Division of liver parenchyma was performed with the LigaSure™/ForceTriad™ platform (Medtronic, Minneapolis, MN, USA) with or without the use of the SonoSurg™ ultrasonic aspirator (Olympus, Tokyo, Japan) and Valleylab™ Argon Beam Coagulator (Medtronic, Minneapolis, MN, USA). Major structures were divided using linear stapler technology (Medtronic, Minneapolis, MN, USA). Total or selective hepatic inflow (Pringle maneuver) was used in selected patients for intervals of maximum 10 minutes. Postoperatively, follow-up was performed with CT of the chest and abdomen every 4 months for the first year and thereafter every 6 months to assess for recurrence.
Sexagenarians as control cohort of patients
Sexagenarians were defined as persons 60–69 years of age. Sexagenarian patients operated in the study period were extracted from the institutional liver database, and half of them randomly selected (n=281) for retrospective update to generate a representative survival curve and comparison to the sexagenarians in the national population.
Age-matched national population
Survival of the age-matched national population of octogenarians (ageM-Octo) was estimated and graphically depicted based on 2010 data available from Statistics Norway (www.ssb.no/en; Table 07902): Alive per. 100,000 inhabitants at: 80 years, 63,728; 81 years, 60,562; 82 years, 57,222; 83 years, 53,791; 84 years, 49,937; 85 years, 46,041; 86 years, 41,963; 87 years, 37,964; 88 years, 33,797; 89 years, 29,599. The data were not adjusted for annual birth rates or net immigration/emigration per year-group. The survival of the age-matched national population of sexagenarian (ageM-Sexa) was estimated similarly.
Statistical analysis
Shapiro-Wilk test was used to assess normal distribution of continuous data allowing expression as the mean with standard deviation (SD) and comparison with independent t-tests. Otherwise, continuous data were expressed as median with range and compared with Mann-Whitney U test. Categorical data were compared by Pearson chi-squared tests. A P value of less than 0.05 was considered statistically significant. Log Rank analyses were used to determine differences between survival curves in Kaplan Meier plots. SPSS v.19.0 (SPSS Inc., IBM, Chicago, IL, USA) was used to perform the statistical analyses.
Results
A total of 59 octogenarians underwent resection of CLM during the study period. The mean age was 82.1 years (SD, 2.1 years) and the median age was 81.6 years (range, 80–89 years); 34 patients (57.6%) were female. The mean observation follow-up time was 42.7 months.
The median number of liver metastases was 1 (range, 1–6) and the mean diameter of the largest metastasis measured before preoperative chemotherapy was 37 mm (SD, 24 mm). The primary tumor lymph node status was negative in 27 (45.8%) patients and positive in 23 (39.0%) patients. The lymph node-status was unknown in 9 (15.3%) patients. The preoperative median CEA was 11 (range, 1–550) ng/mL as listed in Table 1.
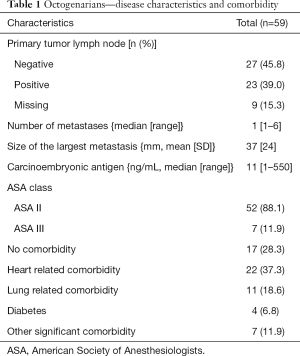
Full table
The patient distribution into the ASA physical status classification system was 52 patients and 7 patients with ASA score II and III, respectively. Heart-related, lung-related, diabetes and other significant comorbidities were registered in 22 patients, 11 patients, 4 patients, and 7 patients, respectively; 17 patients (28.8%) had no comorbidity; 8 patients (13.6%) were smokers at the time of surgery (Table 1).
Twelve patients (20.3%) received preoperative chemotherapy for the liver metastases. One patient initially treated with FLOX and one patient treated with FLIRI were both changed to FLV due to poor tolerance. One patient initially treated with capecitabine was changed to FLOX due to lack of response. Bevacizumab was used together with capecitabine in one patient. No patient received cetuximab or panitumumab. Nine out of 12 patients received less than 6 preoperative cycles. Adjuvant chemotherapy was given to 10 (16.9%) patients and 9 out of 10 patients received less than 6 adjuvant cycles, as outlined in Table 2.
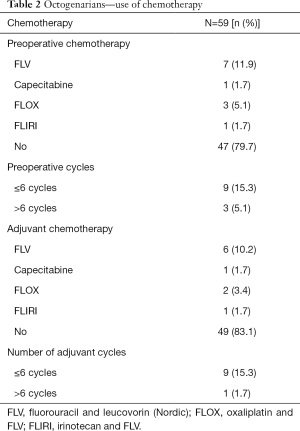
Full table
A positive resection margin (R1) was recorded in 8 patients. The majority of the patients (n=50) underwent minor liver resection and 34 (57.6%) patients were resected laparoscopically. Clavien-Dindo classification of surgical complications postoperatively: grade II, n=4 (blood transfusion); grade IIIa, n=8 (drain placement); grade IIIb, n=3 (reoperation) (14). The 90-day mortality was zero (Table 3).
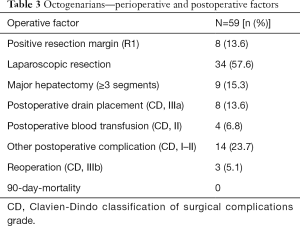
Full table
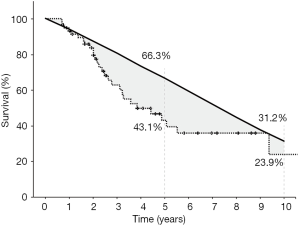
In octogenarians undergoing resection of CLM (n=59), the median OS was 29.6 months. A total of 35 patients died during the observation period, of which 6 died of causes unrelated to colorectal cancer. The 5-year OS was 32.5% compared to estimated 66.3% in the national age-matched population (ageM-Octo); difference 33.8 percentage points (pp). The 10-year OS was 14.1% compared to estimated 31.2% in the age-matched population (ageM-Octo); difference 17.1 pp (Figure 1A).
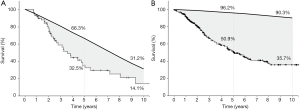
In sexagenarians undergoing resection of CLM (control cohort, n=281), the 5-year OS was 50.9% compared to estimated 96.2% in the national age-matched population (ageM-Sexa); difference 45.3 pp. The 10-year OS was 35.7% compared to estimated 90.3% in the age-matched population (ageM-Sexa); difference 54.6 pp (Figure 1B).
The 5-year and 10-year cancer-specific OS of octogenarians undergoing resection of CLM was 43.1% and 23.9%, respectively (Figure 2). The 5-year RFS of octogenarians undergoing resection of CLM was 32.9% (figure not shown).
Discussion
In the current study, the survival of octogenarians undergoing resection of CLM was compared to the survival of the age-matched national population. This is the first report to incorporate these novel data. A smaller difference between the survival curves was observed between octogenarians and their age-matched population compared to sexagenarians and their age-matched population. This knowledge may be important when assessing octogenarians for liver resection. The relative survival after surgery was poorer in octogenarians, but the benefit of liver resection should be viewed in light of the much lower survival of their age-matched population, thus resection may still be justified.
Interestingly, two years after resection of CLM, the survival of octogenarians was similar to the national age-matched population (ageM-Octo; Figure 1B). Elderly have generally shown reduced tolerance to chemotherapy and in the absence of alternatives, surgery alone may provide oncological control in selected patients. In elderly patients with metastatic colorectal cancer treated with chemotherapy alone, a multicenter experience reported a 2-year survival of 34.8% (15). The data are not directly comparable to the resectable patients in the current study, but the study is interesting: octogenarians selected to chemotherapy also represent a sub-selected group of physical and mentally fit patients compared to those not amenable to neither chemotherapy nor surgery. As such, a 5-year survival of 32.5% may indicate that surgery would be preferable in patients mentally and physically fit to receive either treatment.
Modern approach to patients with CLM includes a multidisciplinary evaluation aimed to uncover biological features of the tumor (16,17). Preoperative chemotherapy is increasingly used to select patients with response or stable disease to surgery (18). In the work-up of elderly, tumor biology may come secondary to physical and mental health (11). In a study of 239 patients undergoing resection of CLM, we previously showed a reciprocal survival course comparing age and recurrence vs. age and survival; young patients (50 years or less) tended to present with recurrence shortly after the liver resection but the recurrence was aggressively treated with re-resections and chemotherapy and they had a superior OS compared to the elderly patients. In contrast, the elderly patients had superior RFS, but inferior OS compared to the younger patients (19). This finding may indicate less aggressive tumor biology in the elderly, a concept that has been investigated in breast cancer, but remains to be fully investigated in colorectal cancer (20-22).
In the current study, preoperative and adjuvant chemotherapy was used in 20.3% and 16.9%, respectively. These rates were low compared to reports on younger patients, and may be explained by the assumption of a lower tolerance of chemotherapy in the elderly. Today, preoperative and adjuvant chemotherapy is used in more than 50% of the patients irrespective of age (19). We did see a trend towards more use of preoperative chemotherapy in the most recent period, also in the elderly (23). However, the elderly patients did primarily receive fluorouracil regimens without the addition of oxaliplatin or irinotecan. Targeted treatment with anti-vascular endothelial growth factor was only used in one patient and anti-epidermal growth factor treatment not used in any.
The primary aim of the current study was to investigate survival in elderly patients compared to the age-matched population. Previous literature has focused on concerns over higher mortality and morbidity rates (3,5,7,24). The surgical management of CLM in elderly patients was examined in a recent large population-based study from Canada (3). The authors defined elderly as patients 75 years or more (n=186), and found a 90-day-mortality rate of 8% despite less aggressive treatment than in younger patients (3). In the current study of 59 octogenarians, the 90-day-mortality was zero, indicating that postoperative mortality may not be an obstacle to liver resection in the elderly. In another study of 41 octogenarians who underwent liver resection, the reported 90-day-mortality was 17.1% while the size and number of tumors were comparable to the current study (5). However, major hepatectomy was performed in 65.9% of the patients compared to 15.3% in the current study of 59 octogenarians. More extensive surgery may have contributed to the higher postoperative mortality rates. As such, a laparoscopic approach and parenchymal sparing techniques utilized in the current study may provide even greater advantages in the elderly (25-27).
Enhanced recovery protocols and fast-track surgery are concepts adapted to improve postoperative health-related quality of life, reduce the length of stay, and lower hospital costs (28,29). In octogenarians the preoperative performance status and level of physical activity (prehabilitation) may be even more important and better predictors of postoperative complications than age (30). The patients of the current study underwent routine examination at the outpatient clinic prior to surgery to determine any comorbidities contraindicating surgery. However, it is uncertain whether a preoperative subjective screening correlates to an objective performance status testing and to postoperative outcome (30). The preoperative assessment and selection of octogenarians to surgery may be improved by implementing objective performance status testing (31). Objective testing opens for the possibility to include elderly patients presenting with poor preoperative performance status in customized training programs to increase level of activity, followed by re-testing of performance status. The concept of prehabilitation could potentially be used for selection purposes in elderly patients where resectability is questionable due to comorbidities. Finally, chronological age may be less important than other selection factors such as physical activity and limited comorbidity (9).
In elderly patients undergoing hepatectomy, complications are the likely cause of loss of independence and mortality, more than the index operation (5). In octogenarians undergoing resection of CLM, a substantial number of patients have already successfully undergone resection of the primary cancer, thus already proven a tolerance for surgery. Furthermore, it is very likely that patients selected for liver resection represent a subgroup of octogenarians healthier than their age-matched population, the cancer set aside. Therefore, a major limitation is the fact that other preoperative selection criteria were not systematically or prospectively recorded, thus difficult to retrieve retrospectively. As successful selection may have contributed to the zero 90-day mortality, this is an important topic worthy of further investigation.
Other limitations of the current study were the fact that octogenarians rejected for surgery (or cause for rejection) were not recorded and, that some caution should be taken when comparing the survival of octogenarians with the estimated survival of the national age-matched population, as the latter was not adjusted for annual birth rates and net immigration and emigration.
A poorer relative OS should not preclude resection of CLM in the octogenarians as the benefit, with respect to survival after surgery, may be similar to that in younger patients. Low levels of postoperative mortality and morbidity was achieved, likely due to selection of mentally and physically fit octogenarians. Two years after surgery, the survival of octogenarian patients was similar to the survival of the national age-matched population. This finding indicate that octogenarians accepted for surgery, regardless of their cancer, had less other life treating conditions than the background population, but also that surgery alone may provide disease control in patients unfit to receive chemotherapy.
Acknowledgements
This study was supported solely by the Department of Hepato-Pancreato-Biliary Surgery, Oslo University Hospital, Oslo, Norway.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the Institutional Review Board (protocol number 2015/18666).
References
- Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet 2014;383:1490-502. [Crossref] [PubMed]
- Brudvik KW, Passot G, Vauthey JN. Colorectal Liver Metastases: A Changing Treatment Landscape. J Oncol Pract 2016;12:40-1. [Crossref] [PubMed]
- Booth CM, Nanji S, Wei X, et al. Management and Outcome of Colorectal Cancer Liver Metastases in Elderly Patients: A Population-Based Study. JAMA Oncol 2015;1:1111-9. [Crossref] [PubMed]
- Cook EJ, Welsh FK, Chandrakumaran K, et al. Resection of colorectal liver metastases in the elderly: does age matter? Colorectal Dis 2012;14:1210-6. [Crossref] [PubMed]
- Shutt TA, Philips P, Scoggins CR, et al. Permanent Loss of Preoperative Independence in Elderly Patients Undergoing Hepatectomy: Key Factor in the Informed Consent Process. J Gastrointest Surg 2016;20:936-44. [Crossref] [PubMed]
- Shirabe K, Kajiyama K, Harimoto N, et al. Early outcome following hepatic resection in patients older than 80 years of age. World J Surg 2009;33:1927-32. [Crossref] [PubMed]
- Leal JN, Sadot E, Gonen M, et al. Operative morbidity and survival following hepatectomy for colorectal liver metastasis in octogenarians: a contemporary case matched series. HPB (Oxford) 2017;19:162-9. [Crossref] [PubMed]
- Adam R, Frilling A, Elias D, et al. Liver resection of colorectal metastases in elderly patients. Br J Surg 2010;97:366-76. [Crossref] [PubMed]
- Cannon RM, Martin RC, Callender GG, et al. Safety and efficacy of hepatectomy for colorectal metastases in the elderly. J Surg Oncol 2011;104:804-8. [Crossref] [PubMed]
- Cho SW, Steel J, Tsung A, et al. Safety of liver resection in the elderly: how important is age? Ann Surg Oncol 2011;18:1088-95. [Crossref] [PubMed]
- de la Fuente SG, Bennett KM, Scarborough JE. Functional status determines postoperative outcomes in elderly patients undergoing hepatic resections. J Surg Oncol 2013;107:865-70. [Crossref] [PubMed]
- Melloul E, Halkic N, Raptis DA, et al. Right hepatectomy in patients over 70 years of age: an analysis of liver function and outcome. World J Surg 2012;36:2161-70. [Crossref] [PubMed]
- Schiergens TS, Stielow C, Schreiber S, et al. Liver resection in the elderly: significance of comorbidities and blood loss. J Gastrointest Surg 2014;18:1161-70. [Crossref] [PubMed]
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13. [Crossref] [PubMed]
- Grande R, Natoli C, Ciancola F, et al. Treatment of Metastatic Colorectal Cancer Patients ≥75 Years Old in Clinical Practice: A Multicenter Analysis. PLoS One 2016;11. [Crossref] [PubMed]
- Poston GJ, Adam R, Alberts S, et al. OncoSurge: a strategy for improving resectability with curative intent in metastatic colorectal cancer. J Clin Oncol 2005;23:7125-34. [Crossref] [PubMed]
- Vauthey JN, Kopetz SE. From multidisciplinary to personalized treatment of colorectal liver metastases: 4 reasons to consider RAS. Cancer 2013;119:4083-5. [Crossref] [PubMed]
- Adam R, de Gramont A, Figueras J, et al. Managing synchronous liver metastases from colorectal cancer: A multidisciplinary international consensus. Cancer Treat Rev 2015;41:729-41. [Crossref] [PubMed]
- Brudvik KW, Bains SJ, Seeberg LT, et al. Aggressive treatment of patients with metastatic colorectal cancer increases survival: a scandinavian single-center experience. HPB Surg 2013;2013. [Crossref] [PubMed]
- Syed BM, Green AR, Paish EC, et al. Biology of primary breast cancer in older women treated by surgery: with correlation with long-term clinical outcome and comparison with their younger counterparts. Br J Cancer 2013;108:1042-51. [Crossref] [PubMed]
- Nardo B, Serafini S, Ruggiero M, et al. Liver resection for metastases from colorectal cancer in very elderly patients: New surgical horizons. Int J Surg 2016;33 Suppl 1:S135-41. [Crossref] [PubMed]
- Shan JL, Li Q, He ZX, et al. A population-based study elicits a reverse correlation between age and overall survival in elderly patients with rectal carcinoma receiving adjuvant chemotherapy. Clin Exp Pharmacol Physiol 2015;42:752-65. [Crossref] [PubMed]
- McCleary NJ, Dotan E, Browner I. Refining the chemotherapy approach for older patients with colon cancer. J Clin Oncol 2014;32:2570-80. [Crossref] [PubMed]
- Tzeng CW, Cooper AB, Vauthey JN, et al. Predictors of morbidity and mortality after hepatectomy in elderly patients: analysis of 7621 NSQIP patients. HPB (Oxford) 2014;16:459-68. [Crossref] [PubMed]
- Mise Y, Aloia TA, Brudvik KW, et al. Parenchymal-sparing Hepatectomy in Colorectal Liver Metastasis Improves Salvageability and Survival. Ann Surg 2016;263:146-52. [Crossref] [PubMed]
- Kokudo N, Tada K, Seki M, et al. Anatomical major resection versus nonanatomical limited resection for liver metastases from colorectal carcinoma. Am J Surg 2001;181:153-9. [Crossref] [PubMed]
- Gold JS, Are C, Kornprat P, et al. Increased use of parenchymal-sparing surgery for bilateral liver metastases from colorectal cancer is associated with improved mortality without change in oncologic outcome: trends in treatment over time in 440 patients. Ann Surg 2008;247:109-17. [Crossref] [PubMed]
- Page AJ, Gani F, Crowley KT, et al. Patient outcomes and provider perceptions following implementation of a standardized perioperative care pathway for open liver resection. Br J Surg 2016;103:564-71. [Crossref] [PubMed]
- Bond-Smith G, Belgaumkar AP, Davidson BR, et al. Enhanced recovery protocols for major upper gastrointestinal, liver and pancreatic surgery. Cochrane Database Syst Rev 2016;2. [PubMed]
- Dunne DF, Jack S, Jones RP, et al. Randomized clinical trial of prehabilitation before planned liver resection. Br J Surg 2016;103:504-12. [Crossref] [PubMed]
- West MA, Asher R, Browning M, et al. Validation of preoperative cardiopulmonary exercise testing-derived variables to predict in-hospital morbidity after major colorectal surgery. Br J Surg 2016;103:744-52. [Crossref] [PubMed]


