Living donor liver transplantation in Europe
Introduction
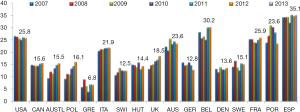
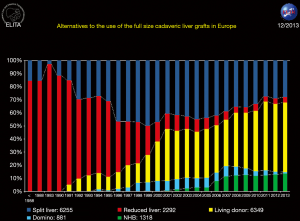
It is difficult to report about living donor liver transplantation (LDLT) in Europe considering the extension and the multi-ethnicity of this wide geographical area with a population of approximately 500 million people in 27 countries with different cultures and different allocation systems (e.g., MELD based system in Euro transplant including Germany, The Netherlands, Belgium, Austria and Slovenia, North Italy, Switzerland and France and centre directed system in UK, Spain, Scandinavian countries). This leads to completely different needs for organs due to different rates of deceased organ donation (Figure 1).
In this context, living organ donation had been introduced by transplant centres as a valuable alternative to bridge the gap between demand and supply of organs. Additionally, the European countries differ significantly for living donation rates, ethical concerns and legislation and protection systems for living organ donors.
In this paper, we will analyse the present European living donor liver activity and highlight the European contribution to the advancement of the field of LDLT. Considering the fact that Turkey is now representing the leading European country in LDLT with more than 1,500 cases during the last 15 years (Tokat Y. Oral communication at ILTS Meeting in San Francisco, USA. 2012), a separate article summarizing the Turkish experience with LDLT can be read in this current issue of HepatoBiliary Surgery and Nutrition (HBSN).
Methods
We first analysed data from European registries (i.e., ELTR, Eurotransplant, EULOD) and from the literature and tried to give a general overview of the actual LDLT activities in Europe. In the second step, we analysed single national registries of different European countries in which LDLT have been performed (when available). Lastly, we reviewed the international literature looking for the peculiarities and contributions given by each country in the field of LDLT. Additionally, it must be mentioned that up to date a LDLT registry is still missing at both European and single national level. The way that the different countries report about their LDLT data is completely different in terms of number and type of items. For these reasons it becomes a very difficult task to achieve wide and complete information about the true LDLT reality in Europe.
European data
LDLT was introduced to Europe in 1991 for children and 1998 for adults (2,3).
From May 1968 to December 2009, the European Liver Transplant Registry (ELTR) collected data concerning 93,634 liver transplantations (LTs) in 83,816 patients from 145 centers of 26 countries (3).
From October 1991 to December 2013, 6,224 LDLTs were performed in 78 out of 155 European LT-centers (4). Figure 2 represented the chronological evolution of LDLT in Europe (4).
The distribution of LDLT according to the recipient age was as follows (4): 1,266 (0−2 years); 8,66 (2−15 years); 1,362 (15−45 years); 2,006 (45−60 years) and 724 (>60 years). In summary, in the recent 25 years, 2,132 paediatric and 4,092 adult LDLT procedures have been performed respectively.
According to the recent ELTR report by Adam et al. who analysed the overall LT activity in Europe till 2009, the most active European countries performing LDLT were: Turkey (n=1,217), Germany (n=715), France (n=409), Belgium (n=341), Italy (n=212), Spain (n=189), United Kingdom (n=138) and Poland (n=135) (4). However, large differences across European countries in the rates of living donation could be observed. Living liver donations were more frequent in the North-Western region than in the Eastern and Mediterranean ones. Possible barriers for increasing living donation were financial barriers, a negative attitude among health care professionals towards such programmes and lack of surgical expertise (6,7).
In 2011, 6,808 patients were on waiting lists for LT in 27 European Union (EU) countries (502 million inhabitants), while during the same year 7,006 LTs were performed, 3.5% (0−50%) of which were LDLTs (7,8).
According to ELTR data from 1991 to 2013, 7 cases of donor mortality have been reported (overall donor surgical mortality rate 0.18%), but only 6 have been officially reported in the literature (see Table 1). At this regard we can reinforce the importance of an accurate and extensive preoperative assessment of the donor in order to avoid future mortality events (9,12).

Full table
Overall, 1-, 5-, 10- and 15-year graft survival rate after LDLT were 80%, 69%, 61% and 60% respectively. The results were better in children than in adults (4,13). Graft survival rate after LDLT was better than that after DDLT in children (78% vs. 72%, P<0.001), but similar in adults (64% vs. 63%) (4,13).
Overall, graft loss after LDLT included more technical complications (26% vs. 14%), more infections (23% vs. 18%), more rejections (8% vs. 4%), and more tumour recurrence (12% vs. 9%), but less general complications (20% vs. 27%) and less non-tumor disease recurrence (4% vs. 12%) than that after DDLT (all P<0.05) (3,4). The reason for higher tumour recurrence rates in LDLT recipients may be the fact that LDLT recipients exceeded the Milan or even the UCSF criteria more often than the DDLT ones did (14-16).
Eurotransplant (ET) (www.eurotransplant.org)
Eight European countries are actually members of ET organisation: Austria, Belgium, Croatia, Germany, Hungary, Luxembourg, Netherlands and Slovenia. All together these Countries have a population of 134 million people served globally by 41 LT centres.
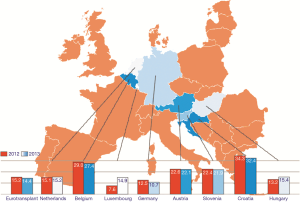
Organ sources are deceased donor (DD) (DCD, donor after cardiac death, only in B, NL and A) and LD. Figure 3 reported the numbers of DD per million population in each ET country in 2012 and 2013. A very high discrepancy among different ET countries in terms of DD rates can be observed. Austria, Belgium, Croatia and Slovenia are leading countries and Germany is the worst one in terms of DDs per million people (pmp).
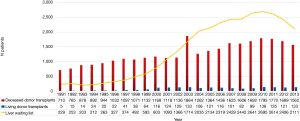
In 2013, 2,041 patients were waiting for a LT (more than 40% of them waiting longer than 2 years), 1,695 were transplanted and 487 died on waiting list (37% of them with MELD scores >30). Almost 55% of DDLT were performed in recipients with MELD >25 (i.e., less than 10% of all patients on active waiting list).
Therefore, for the remnant 90% of patients who are also in need of a LT, a solution is urgently needed. LDLT could be one possible solution. Unfortunately, according to data reported in Figure 4, it is evident that LDLT still represents an under-utilized source of organs in ET area (e.g., in ET 2011, 2012 and 2013, 119, 116 and 130 LDLTs have been respectively performed, i.e., 7.4%, 6.6% and 7.8% of all LT activity in this area). Germany and Belgium are the leading countries in ET area concerning the LDLT activity, in 2013, 64% (86/133) in Germany and 31% (42/133) in Belgium (17).
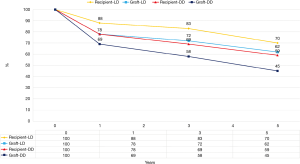
Unfortunately, with exception of patients and graft survival rates (see Figure 5) in ET-database no further results like donor morbidity and mortality have been reported.
Germany (80 million inhabitants, 10.7 DD pmp)
The actual situation of LT in Germany has reached in the recent years dramatic aspects. The DD rates are progressively decreasing during the last 3 years in 2013 (10.9 DD pmp) (18). Additionally, over the last 10−15 years the quality of donor organs has seen a continuous deterioration. A total of 63% of organs are “suboptimal” with a donor risk index almost constantly >1.5 (19). The consequence is a progressively increasing need of liver grafts. In 2013, 1,534 patients were actively listed for LT, but only 970 could be transplanted.
On top of that, the absence of a national split policy and the introduction of a MELD based allocation system further worsened the situation. In fact, in Germany incentives for transplant centres are inappropriate. Patients with decompensated cirrhosis, high MELD scores and high post-transplant mortality as well as marginal liver grafts are accepted for transplantation without the necessary consideration for outcomes. Moreover the transplant centers have no regulatory mandate for making public their results (19).
The matched MELD scores of the recipients who are currently transplanted have progressively increased. Although at the beginning of the MELD-system in 2007, donor livers were allocated to the mean match MELD score of 25, in 2010 it increased up to 34. Weissmüller et al. reported poor survival rates in patients with a lab MELD >30 around 55% at 1 year (20-22).
Thus, the small children and adult with MELD scores <25 who do not benefit from the actual MELD allocation system should be the best candidates for LDLT in Germany (21).
Unfortunately, at the moment LDLT represents an under-utilized resource of liver grafts (see Figure 6). According to the German Organ Procurement Organization (i.e., DSO) database in 2011, 2012 and 2013, the ratios between LDLT and total LT performed in Germany were 5% (71/1,199), 7% (78/1,097) and 8% (83/970) respectively (18). The reason is unclear and subjected to speculative explanations like scepticisms and cultural attitude against LD, missing a clear, adequate and honest information about the procedure and its related complications, and lack of dedicated highly specialized personnel in form of trained interdisciplinary teams (not only surgeon). Last but not least, potential living donors still may not feel themselves protected enough by the system from any kind of complications (medical, psychological and occupational).
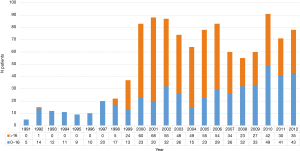
Actually, only 11 of 24 German LT centres, are performing LDLT actively and among them, the main representatives are Regensburg and Essen (mainly paediatric LDLT dedicated), Hannover (totally LDLT paediatric dedicated) and Jena (totally AALDLT dedicated) (see Table 2).
Interestingly, there are very few reports about donor and recipient results in LDLT in Germany. Recently, Settmacher et al. (21) descriptively analysed the data of Adult to Adult LDLT in Germany from 2007 to 2009 by using a multi-center retrospective analysis via a questionnaire and data provided by ET. Ten German centers performed 84 LDLTs in adults, ranging from 1 to 16 AALDLT per center. HCC with cirrhosis (15/84) was the most common indication of LDLT. The recipient mean lab-MELD score was 15±8. Six re-transplantations were required after initial LDLT. The 1-year patient survival rate was 81%. The incidence of complications in LD was 30.4% (n=24). Biliary complications occurred in 7 LD and 1 of them had a surgical repair. All together 5 patients were re-operated (i.e., Dindo-Clavien grade III complication). No donor mortality was reported in the observation period (21).
Despite the present little activity, some relevant contributions have been brought by the German group led by Broelsch & Malago in the Hamburg and Essen transplant centres. The milestones of this school can be summarized as following:
- Development and establishment of the first LDLT program in Germany and one of the first in Europe (2,23-25);
- Introduction and development of AALDLT in Germany and Europe (first AALDLT 1998, especially right AALDLT) (2,26,27);
- Essential imaging and anatomy studies of the liver in the settings of segmental LT which influenced the surgical strategy of donor hepatectomy (28-34);
- Although there is no German-wide common imaging policy, most of German LDLT centers perform nowadays anatomy studies of the liver by means of Angio-CT (for vascular anatomy) and MRI scans for biliary anatomy. Most of centres do additionally perform a 3D reconstruction of vascular and biliary anatomy by MeVis Bremen (9,12);
- Reports about quantitative functional and volumetric liver regeneration after liver donation (35,36);
- Particular care of donor safety in its different aspects. In particular, donor’s complications were reported honestly and this school was the first to (and until now the only one) introduce the classification of psychological complication after living donation (9,37,38);
- Relevant contribution in the field of psychosomatics aspects of living liver donors (39-42).
Belgium (11.2 million inhabitants, 27 DD pmp)
According to the ELTR data, Belgium is one of the European leading countries in LDLT. Unfortunately, very few data are available online the national Belgian transplant registry with exception of the data presented at the 18th annual meeting of the Belgian Transplantation Society (BTS) held in March 2011 in Brussels (43).
In the period 2000−2010, a total of 279 LDLTs were performed in Belgium with a median value of 25 LDLT per year (range, 13−40). The chronological distribution of LDLT performed in Belgium in this period is reported in Figure 7.
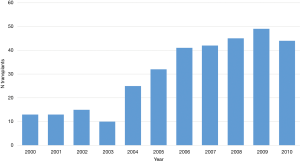
In the period 1999−2010, 136 AALDLT were performed in 4 Belgian LT-centers: University Hospital Gent (n=81), University Hospital Saint-Luc-Woluwe in Brussels (n=30), University Hospital CHU-Sart Tilman in Liege (n=19) and Catholic University Hospital in Leuven (n=6) (courtesy of Prof Troisi, Gent). Overall, recorded donor’s complications in Belgium are comparable to those reported in the European Registry. Specific complications are mainly represented by biliary fistulas (7%). Forty-eight donors experienced one or more complications with 11 donors suffering a complication greater than 3 according to the Dindo Clavien classification. One donor death was also reported.
Two Belgian centers marked the landscape of LDLT in a relevant way.
The LT-center of Catholic University St Luc in Brussels has recently reported their single center experience with 250 pediatric LDLTs from 1993 till 2012 (almost 80% of all pediatric LT from 2009−2012) which does represent one of the single centre experience in Europe (44).
The short and long term results are excellent with 1-, 5-, and 10-year patient and graft survival rates as 96.0%, 93.9%, 93.2%, and 95%, 91% and 90% respectively.
The same group introduced the particular innovative technique of portoplasty in case of hypoplasia of portal vein. In biliary atresia recipients, the rate of PV complications was lower after portoplasty (4.6%) than after truncal PV anastomosis (9.8%) and jump graft interposition (26.9%; P=0.027) (44).
Interestingly, the same Authors observed that maternal donation might be a protective factor for acute rejection in pediatric LDLT (44).
The LT-center of the University of Gent provided important milestones in the evolution of AALDLT:
- They were the first to perform adult to adult LDLT with right lobe in Belgium (45);
- Propagated the left lobe for AALDLT;
- Introduced and the laparoscopic donor operation (initially only left lateral, followed by full left and finally right) (46,47). Troisi et al. have showed very recently that the laparoscopic donor hepatectomy is associated with intraoperative results and surgical outcomes that are comparable to results of the open procedure, but donor morbidity rates and wound-related complications are significantly reduced in the laparoscopic procedure (46-49);
- Provided essential concepts in management of small for size syndrome through accurate studies of liver haemodynamic and introduced systematic concepts and strategy of graft inflow modulation (50-52);
- Developed novel immunosuppressive protocols allowing withdrawal or minimization of immunosuppressive therapy after transplantation with donor stem cell infusion (53);
- Introduced one of the first protocols of ABO incompatible LDLT in Europe (54).
Italy (59.8 million inhabitants; 21.9 DD pmp)
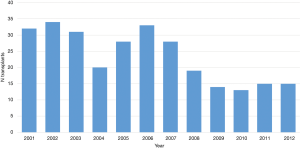
According to the data reported by Italian National Transplant Center (55) from 2001 to 2013, 15 Italian LT centers performed a total number of 282 LDLTs. The chronological distribution is reported in Figure 8. At this regard, Pretagostini et al. observed that after enthusiastic adoption in the1990s, LDLT rates dramatically decreased in recent years: from 34 in 2002 (3.9% of total LT) to 13 in 2010 (1.3% of total LT) (56).
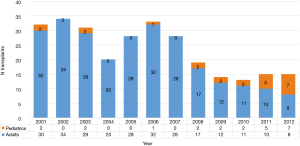
In the period of 2001−2013, leading Italian centers in the field of LDLT were those in Palermo and Milano Niguarda with a total number of 107 and 80 LDLT procedures each. Actually active LDLT centers are Milano Niguarda, Palermo ISMETT and Bambin Gesú Rome (the first one performing exclusively AALDT and the last one exclusively pediatric LDLT, while Palermo performs both).
Interestingly, opposite to most of other European countries, LDLT in Italy has been mainly adopted for adult recipients (Figure 9). Pediatric LDLT in Italy accounts for almost 12% of all LDLT activities. This is probably due to an efficient mandatory split policy in this country which makes DDLT become the main source of liver grafts for children (57). At “Bambin Gesú Children Hospital” in Rome since 2011 to present, 35 pediatric LDLTs have been performed, with 100%, patient and graft survival rate (personal communication by J. de Ville de Goyet).
The reported 1-, 2-, 4- and 6-years overall patient and graft survival rate were 85%, 79%, 70% and 68%, and 80%, 72%, 68% and 65% respectively (55).
Unfortunately, no data about donor morbidity and mortality have been reported.
The Italian data on LDLT reported in the international literature showed that only 2 centers (Palermo and Milan) reported about their single center experience.
At the “Istituto Mediterraneo per i Trapianti e Terapie ad Alta Specializzazione” in Palermo, from January 2002 to April 2013, 107 LDLTs were performed (95 for adult patients and 12 for pediatric recipients) (58). No living donor mortality was reported; 25 (27.1%) living donors presented one or more complications in the post-operative period. The patient and graft survival rate at 1-, 3- and 5-year after right lobe LDLT were 89.3%, 83.2%, 77.8% and 83.4%, 77.3%, 71.9% respectively. From technical point of view, it must be mentioned that this center routineously performs laparoscopic left lateral segmentectomy for pediatric recipient and has been the first in the Europe to perform an entirely robotic living donor right hepatectomy (58).
At the center of Milano Niguarda, from 2001 to 2012, 80 AALDT with right lobes without MHV were performed. With a median follow up of 63.2±12.6 months, the donor mortality was 0%. Two donors experienced intraoperative complications, but both of them had completely recovered thereafter. Among the 22 complications in 17 donors (21.2%), 7 (8.7%) were major complications (Clavien grade ≥ 2b) but only 2 donors required surgical treatment. Unfortunately the Authors did not report their results of graft and recipient survival (59).
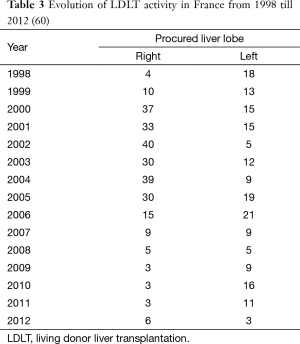
France (66.03 million inhabitants, 25.9 DD pmp)
According to the data from the French Agency of Biomedicine (60) in France, only 7 out of 24 LT centers do perform LDLT (four centers in Paris, Lyon, Nice and Rennes). The Hôpital Paul Brousse in Villejuif is the one with the most experience in right lobe LDLT (61). In France, from 1998 until December 2014, 22,204 LTs and 465 LDLTs have been performed. Since 2007, a significant decrease of LDLT activity has been observed (see Table 3). For example, from 2010 till 2012, a total of only 40 LDLTs were performed in France (20 adult and 20 pediatric LDLT respectively) accounting for less than 1% of whole LT activities (60).
The patient survival rates after LDLT at 1-, 5- and 10-year are respectively 86%, 78% and 72% (60).
The main indication of LDLT in children is represented by cholestatic liver disease. The paediatric patient and graft survival rates at 5 years are 83% and 77% respectively. In case of severe acute liver failure, the survival rate at 5 years is only 68%.
In adults, the main indication of LDLT is represented by HCC. The 5-year patient and survival rates are 61% and 52% respectively. Interestingly, it seems that the survival rates after AALDLT depend on the volume of liver graft (i.e., 75% for right lobe and only 40% for left lobe). The postoperative mortality has been reported to be similar to the post DDLT one (i.e., 12.5%) (60).
Being opposite to other national registries, the national French agency is the only one reporting data about donor morbidity and mortality. Up to now, two donor’s deaths have been reported (Lyon and Paris). The global mortality risk for the LD is reported to be 0.27% to 0.5% after right lobe donation and 0.1% after left lobe donation.
The donor morbidity rates are varying between 18% and 60% (60,61). Also, in this case, the rates of complications seem to be proportional to the amount of resected liver volume.
From the technical point of view, the French surgeons provided important contribution in the field of LDLT:
- Boillot et al. introduced 2002 new concepts of graft inflow modulation in the form of meso-caval shunt as prevention of small for size syndrome (62);
- Development of the laparoscopic donor’s hepatectomy (63). Recently, Scatton et al. have reported about their single center experience with pure laparoscopic left lateral hepatectomy in 64 LDs (64). Eleven donors (16%) had grade 1 complications, according to the Dindo-Clavien classification. Five donors (7.1%) presented grade 2 complications, and 1 donor (1.4%) had a grade 3 complications. No donor death occurred. Overall, patient and graft survival rates for pediatric recipients were 95% and 92% at 1 year, 95% and 88% at 3 years, and 95% and 84% at 5 years, respectively. This made the laparoscopic retrieval of the left lateral section for LDLT become a safe, reproducible and established procedure in France (64);
- Introduction and development of AALDLT with left lobe grafts (65).
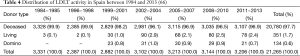
Spain (46.9 million inhabitants, 35 DD pmp)
According to the Spanish national transplant registry [“Sociedad Española de Trasplante Hepatico, SETH” (66)] from 1984 till 2013 a total of 351 LDLTs were performed: 180 ped, LDLTs (54%) and 155 AALDLTs (46%). LDLT represents almost 2.5% of whole Spanish LT activity (0.1−2.9%) (see Table 4). This may be due to the high rates of organ donation from DD. Although 8 out of 26 LT centres have historically performed LDLT in Spain, at present, only 5 centres are active: 3 for pediatric LDLT (La Paz Infantil and 12 de Octubre Infantil in Madrid and Val d’Hebron Infantil in Barcelona) and 2 for adult LDLT (Clinic Provincial in Barcelona and Clinica Universidad de Navarra in Pamplona).
Leading centres in LDLT in Spain are represented by Hospital La Paz in Madrid, Hospital Clinic i Provincial in Barcelona and Hospital 12 de Octubre in Madrid with 130, 91, and 53 LDLT procedures each.
The reported graft survival rates after AALDLT at 1, 3, 5 and 10 years were 81%, 73%, 67% and 55% respectively. The graft survival rates after PLDLT at 1, 3, 5 and 10 years were 82%, 81%, 76% and 74% respectively (66).
Different from DDLT, the international literature is poor of Spanish reports about LDLT. Nonetheless two centers marked the landscape of LDLT from scientific and technical point of views:
- The Hospital Clinic from Barcelona reported interesting and innovative results about:
- Experimental and clinical results of haemodynamic management of SFSS after LDLT (67-70);
- Recurrence of hepatitis C after LDLT (71);
- Improvement of imaging of biliary anatomy with MRI (72).
- The Clinica Universidad de Navarra also played an important role in the development of following aspects of LDLT:
United Kingdom (64 million inhabitants, 18.5 DD pmp)
According to the UK national transplant registry in UK, 6 out of 7 LT centres have performed LDLT but the majority of LDLTs are currently performed in only three centres: King’s College Hospital in London, Queen Elizabeth Hospital in Birmingham and St James’s University Hospital in Leeds. Among them, the King’s College is the leading centre in UK performing more than 60% of all LDLTs in UK in the period from 2012−2013 (76-78).
Table 5 reported the yearly distribution of LDLT activity in UK in both children and adult recipients between 2008 and 2014. It can be noticed that LDLT does represent less than 4% of overall LT activities.
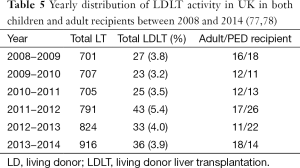
Full table
Unfortunately, the national UK registry does not provide further data regarding donor and recipient results.
Dettani et al. reported about 50 paediatric LDLTs performed at King’s College in London from 1993 to 2008. Patient and graft survival rates were 97.8%, 95.1% and 95.1%, and 97.8%, 92.1% and 71.7% at 1, 5 and 10 years post-transplant respectively (79).
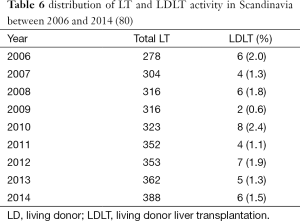
Scandinavia (25 million inhabitants, 13.6−15.1 DD
pmp)
Countries members of Scandiatranplant are: Norway, Denmark, Sweden, Finland and Iceland.
According to data from the Nordic liver transplant registry (80), LDLT in this area is a very under-represented reality (i.e., less than 2% of all LT activity) (see Table 6).
Actually, LDLT is performed only in two Swedish centres, which are Gothenburg and Stockholm. Gothenburg is the first centre which performed LDLT in Scandinavia (81).
Similarly to UK registry, no data about donor and recipients results have been provided.
In the international literature we found a single centre report about seven paediatric LDLTs performed in Stockholm from 1995 to 2003. The Authors reported excellent results, but also in this case no further data were provided for both donor and recipients (82).
Olausson et al. from Gothenburg recently have reported extreme innovative and original application of LDLT: auxiliary LDLT in highly immunized highly sensitized LD kidney transplant recipients as a sort of biological in vivo immune absorbing column. In this context, the liver protects the kidney from the harmful HLA antibodies. Seven patients, with broadly reacting HLA antibodies and positive crossmatches, were successfully transplanted with a partial liver and a kidney from the same donor (83).
Poland (38.2 million inhabitants, 16.1 DD pmp)
According to the national Polish transplant registry (84) in 2014, 2013 and 2012, 336, 318 and 314 LTs, as well as 30, 18 and 14 LDLTs (i.e., 9%, 5% and 4% of whole LT activity) have been performed respectively.
Unfortunately, no data about donor and recipient results have been reported.
The leading Polish centre is the Warsaw University Transplant Center where 120 paediatric LDLTs were performed (109 left lateral grafts and 11 grafts consisting of segments II, III, and IV) between October 1999 and January 2008 (85).
Switzerland
In view of the permanent shortage of organs in Switzerland, the AALDLT was started in 1999 in Geneva University Hospitals (86).
According to data from Swiss transplant registry in 2013, 305 patients were waiting for a LT, and 109 LT (of which 5 LDLTs) were performed in the 3 Swiss LT centres (Bern, Geneva and Zurich).
No further data about donor and recipient results have been reported.
Others
Last but not least, we would like to mention the recent activities and projects promoted by the European Union in terms of living organ donation in Europe.
EULOD project
The project on Living Organ Donation in Europe (EULOD) started in March 2010 and ended in September 2012. It aimed to establish an inventory of living donation practices in Europe, explore and promote living donation as a way to increase organ availability, and to present recommendations to improve the quality and safety of living organ donations in Europe. The results have been reported in the book: “The EULOD Project: Results and Recommendations” (87).
EULID (Euro Living Donor) project
EULID is a project co-funded by the European Union in the framework of the EU Health Programme 2003−2008, and its main objective was to analyse the current European situation regarding legal, ethical, protection and registration practices related to living organ donation, in order to set standards and recommendations that guarantee the living donor health and safety (88).
ELIPSY (Euro Living Donor Psychosocial Follow-up) project
The main objective of the ELIPSY project is to contribute to guarantee the good quality of organ living donation for transplant through a living donor long-term psychosocial and quality of life follow-up and also to correlate those aspects with the recipient’s outcome with the creation of a follow-up methodology. That will contribute to guarantee health and safety in psychosocial terms of living donors with the contribution to harmonize the proceedings in Europe (89).
Conclusions
From this analysis of LDLT in Europe, it is clear that this transplant modality has not been wholeheartedly embraced and in some countries, it has been completely abandoned. The reasons are multifactorial but they are probably similar to the reasons that have made LDLT lose favour in USA. Donor risk, technical challenges both in the donor and the recipient operation, introduction of the MELD score, increased utilization of marginal donors seem to be the most possible causes of the decline of LDLT in Europe. Nonetheless, the contributions that the European Centres have made to the advancement of LDLT are many and significant, spanning from the development of sophisticated 3D imaging, to reconstruction of the donor liver, surgical solutions to venous outflow and portal inflow of the transplanted graft, studies regarding the outcome improvements in small for size LT and last but not least, the recent development of laparoscopic donor hepatectomy. Moreover, although the sheer number of living donor liver transplant performed is not large, the interest for the procedure from the transplantation government bodies is still high and the need of the patients awaiting LT is unchanged. Therefore, it is possible that this decline is temporary and a renewed interest in the procedure will occur.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Spanisch National Transplant Organisation - Organización Nacional de Trasplantes. Annual rates of deceased donors pmp in Europe from 2005-2013. Available online: http://www.ont.es/infesp/Paginas/DatosdeDonacionyTrasplante.aspx
- Broelsch CE, Malago M, Testa G, et al. Living donor liver transplantation in adults: outcome in Europe. Liver Transpl 2000;6:S64-5. [PubMed]
- Dutkowski P, De Rougemont O, Müllhaupt B, et al. Current and future trends in liver transplantation in Europe. Gastroenterology 2010;138:802-9.e1-4.
- European Liver Transplant Registry. Documents to download. Available online: http://www.eltr.org/spip.php?page=documents
- Registry ELT. Evolution of LTs in Europe European Liver Transplant Registry. Available online: http://www.eltr.org/Evolution-of-LTs-in-Europe.html
- Lennerling A, Lovén C, Dobbels F, et al. Living Organ Donation in Europe-Clinical Praxis. In: Ambagtsheer A, Weimar W, editors. The EULOD Project Living Organ Donation in Europe Results and Recommendations. D-49525 Lengerich: Pabst Science Publishers, 2013:9-26.
- Lennerling A, Loven C, Dor FJ, et al. Living organ donation practices in Europe - results from an online survey. Transpl Int 2013;26:145-53. [PubMed]
- Matesanz R. International figures on donation and transplantation 2011, Council of Europe. Newsletter Transplant 2012;17.
- Nadalin S, Malago M, Radtke A, et al. Current trends in live liver donation. Transpl Int 2007;20:312-30. [PubMed]
- Melloul E, Dondero F, Paugam-Burtz C, et al. Living liver donor death related to complications of myeloma. Liver Transpl 2009;15:326-9. [PubMed]
- Detry O, De Roover A, Lauwick S, et al. Fatal small for size syndrome after right lobe donation. Transplant Int 2011;24:8.
- Nadalin S, Capobianco I, Konigsrainer I, et al. Living liver donor: indications and technical aspects. Chirurg 2015;86:609-21. [PubMed]
- Adam R, Karam V, Delvart V, et al. Evolution of indications and results of liver transplantation in Europe. A report from the European Liver Transplant Registry (ELTR). J Hepatol 2012;57:675-88. [PubMed]
- Malagó M, Sotiropoulos GC, Nadalin S, et al. Living donor liver transplantation for hepatocellular carcinoma: a single-center preliminary report. Liver Transpl 2006;12:934-40. [PubMed]
- Park MS, Lee KW, Suh SW, et al. Living-donor liver transplantation associated with higher incidence of hepatocellular carcinoma recurrence than deceased-donor liver transplantation. Transplantation 2014;97:71-7. [PubMed]
- Fouzas I, Sotiropoulos GC, Lang H, et al. Living donor liver transplantation for hepatocellular carcinoma in patients exceeding the UCSF criteria. Transplant Proc 2008;40:3185-8. [PubMed]
- Eurotransplant. Annual Report 2013. Available online: http://www.eurotransplant.org/cms/mediaobject.php?file=AR20135.pdf
- German Organ Transplantation Foundation. Registry. Available online: http://www.dso.de/servicecenter/downloads/jahresberichte-und-grafiken.html
- Schlitt HJ, Loss M, Scherer MN, et al. Current developments in liver transplantation in Germany: MELD-based organ allocation and incentives for transplant centres. Z Gastroenterol 2011;49:30-8. [PubMed]
- Berg CL, Merion RM, Shearon TH, et al. Liver transplant recipient survival benefit with living donation in the model for endstage liver disease allocation era. Hepatology 2011;54:1313-21. [PubMed]
- Settmacher U, Gotz M, Rahmel A, et al. Living donor liver transplantation in adults in the MELD era in Germany--a multi-center retrospective analysis. Transpl Int 2011;24:904-11. [PubMed]
- Weismüller TJ, Fikatas P, Schmidt J, et al. Multicentric evaluation of model for end-stage liver disease-based allocation and survival after liver transplantation in Germany--limitations of the ‘sickest first’-concept. Transpl Int 2011;24:91-9. [PubMed]
- Broelsch CE, Burdelski M, Rogiers X, et al. Living donor for liver transplantation. Hepatology 1994;20:49S-55S. [PubMed]
- Malagó M, Rogiers X, Burdelski M, et al. Living related liver transplantation: 36 cases at the University of Hamburg. Transplant Proc 1994;26:3620-1. [PubMed]
- Wadström J, Rogiers X, Malago M, et al. Experience from the first 30 living related liver transplant in Hamburg. Transplant Proc 1995;27:1173. [PubMed]
- Broelsch CE, Frilling A, Testa G, et al. Living donor liver transplantation in adults. Eur J Gastroenterol Hepatol 2003;15:3-6. [PubMed]
- Malagó M, Testa G, Frilling A, et al. Right living donor liver transplantation: an option for adult patients: single institution experience with 74 patients. Ann Surg 2003;238:853-62. [PubMed]
- Radtke A, Nadalin S, Sotiropoulos GC, et al. Computer-assisted operative planning in adult living donor liver transplantation: a new way to resolve the dilemma of the middle hepatic vein. World J Surg 2007;31:175-85. [PubMed]
- Radtke A, Sgourakis G, Molmenti EP, et al. Computer-assisted surgical planning in adult-to-adult live donor liver transplantation: how much does it help? A single center experience. Transplantation 2012;94:1138-44. [PubMed]
- Radtke A, Sgourakis G, Sotiropoulos GC, et al. Donor/recipient algorithm for management of the middle hepatic vein in right graft live donor liver transplantation. Am J Surg 2010;199:708-15. [PubMed]
- Radtke A, Sgourakis G, Sotiropoulos GC, et al. Territorial belonging of the middle hepatic vein in living liver donor candidates evaluated by three-dimensional computed tomographic reconstruction and virtual liver resection. Br J Surg 2009;96:206-13. [PubMed]
- Radtke A, Sotiropoulos GC, Molmenti EP, et al. Transhilar passage in right graft live donor liver transplantation: intrahilar anatomy and its impact on operative strategy. Am J Transplant 2012;12:718-27. [PubMed]
- Schroeder T, Malago M, Debatin JF, et al. “All-in-one” imaging protocols for the evaluation of potential living liver donors: comparison of magnetic resonance imaging and multidetector computed tomography. Liver Transpl 2005;11:776-87. [PubMed]
- Schroeder T, Nadalin S, Stattaus J, et al. Potential living liver donors: evaluation with an all-in-one protocol with multi-detector row CT. Radiology 2002;224:586-91. [PubMed]
- Jochum C, Beste M, Penndorf V, et al. Quantitative liver function tests in donors and recipients of living donor liver transplantation. Liver Transpl 2006;12:544-9. [PubMed]
- Nadalin S, Testa G, Malago M, et al. Volumetric and functional recovery of the liver after right hepatectomy for living donation. Liver Transpl 2004;10:1024-9. [PubMed]
- Nadalin S, Malago M, Testa G, et al. “Hepar divisum”--as a rare donor complication after intraoperative mortality of the recipient of an intended living donor liver transplantation. Liver Transpl 2006;12:428-34. [PubMed]
- Valentín-Gamazo C, Malagó M, Karliova M, et al. Experience after the evaluation of 700 potential donors for living donor liver transplantation in a single center. Liver Transpl 2004;10:1087-96. [PubMed]
- Erim Y, Beckmann M, Kroencke S, et al. Sense of coherence and social support predict living liver donors’ emotional stress prior to living-donor liver transplantation. Clin Transplant 2008;22:273-80. [PubMed]
- Erim Y, Beckmann M, Kroencke S, et al. Influence of kinship on donors’ mental burden in living donor liver transplantation. Liver Transpl 2012;18:901-6. [PubMed]
- Erim Y, Beckmann M, Kroencke S, et al. Psychological strain in urgent indications for living donor liver transplantation. Liver Transpl 2007;13:886-95. [PubMed]
- Erim Y, Beckmann M, Valentin-Gamazo C, et al. Quality of life and psychiatric complications after adult living donor liver transplantation. Liver Transpl 2006;12:1782-90. [PubMed]
- Troisi RI, Bosmans JL. Organ donation in Belgium 2010: an increase of combined organ transplants and living donor liver transplantations. Transplant Proc 2011;43:3439-40. [PubMed]
- Gurevich M, Guy-Viterbo V, Janssen M, et al. Living Donor Liver Transplantation in Children: Surgical and Immunological Results in 250 Recipients at Universite Catholique de Louvain. Ann Surg 2015;262:1141-9. [PubMed]
- Troisi R, Cuomo O, De Hemptinne B. Adult-to-adult living-related liver transplantation using the right lobe. Case report. Digestive and liver disease 2000;32:238-42. [PubMed]
- Troisi RI. Open or laparoscopic living donor liver hepatectomy: still a challenging operation! Am J Transplant 2014;14:736. [PubMed]
- Troisi RI, Wojcicki M, Tomassini F, et al. Pure laparoscopic full-left living donor hepatectomy for calculated small-for-size LDLT in adults: proof of concept. Am J Transplant 2013;13:2472-8. [PubMed]
- Berardi G, Tomassini F, Troisi RI. Comparison between minimally invasive and open living donor hepatectomy: A systematic review and meta-analysis. Liver Transpl 2015;21:738-52. [PubMed]
- Troisi R, Debruyne R, Rogiers X. Laparoscopic living donor hepatectomy for pediatric liver transplantation. Acta Chir Belg 2009;109:559-62. [PubMed]
- Sainz-Barriga M, Reyntjens K, Costa MG, et al. Prospective evaluation of intraoperative hemodynamics in liver transplantation with whole, partial and DCD grafts. Am J Transplant 2010;10:1850-60. [PubMed]
- Sainz-Barriga M, Scudeller L, Costa MG, et al. Lack of a correlation between portal vein flow and pressure: toward a shared interpretation of hemodynamic stress governing inflow modulation in liver transplantation. Liver Transpl 2011;17:836-48. [PubMed]
- Troisi R, Cammu G, Militerno G, et al. Modulation of portal graft inflow: a necessity in adult living-donor liver transplantation? Ann Surg 2003;237:429-36. [PubMed]
- Donckier V, Troisi R, Le Moine A, et al. Early immunosuppression withdrawal after living donor liver transplantation and donor stem cell infusion. Liver Transpl 2006;12:1523-8. [PubMed]
- Troisi R, Noens L, Montalti R, et al. ABO-mismatch adult living donor liver transplantation using antigen-specific immunoadsorption and quadruple immunosuppression without splenectomy. Liver Transpl 2006;12:1412-7. [PubMed]
- Italian Ministry of Health. Statistics. Available online: http://www.trapianti.salute.gov.it/cnt/cntStatistiche.jsp?area=cnt-generale&menu=menuPrincipale&label=datdoc&titolo=1111&etichetta=
- Pretagostini R, Ricci A, Gabbrielli F, et al. Living organ donation, a therapeutic possibility, is still poorly used in Italy: a national analysis. Transplant Proc 2012;44:1818-9. [PubMed]
- Gridelli B, Spada M, Petz W, et al. Split-liver transplantation eliminates the need for living-donor liver transplantation in children with end-stage cholestatic liver disease. Transplantation 2003;75:1197-203. [PubMed]
- Gruttadauria S, Pagano D, Cintorino D, et al. Right hepatic lobe living donation: a 12 years single Italian center experience. World J Gastroenterol 2013;19:6353-9. [PubMed]
- Lauterio A, Poli C, Cusumano C, et al. Living-donor liver transplantation: donor selection criteria and postoperative outcomes. A single-center experience with a 10-year follow-up. Transplant Proc 2013;45:2680-3. [PubMed]
- French Agency of Biomedicine. The medical and scientific report of the Agency of Biomedicine 2013. Available online: http://www.agence-biomedecine.fr/annexes/bilan2013/donnees/organes/05-foie/synthese.htm
- Azoulay D, Bhangui P, Andreani P, et al. Short- and long-term donor morbidity in right lobe living donor liver transplantation: 91 consecutive cases in a European Center. Am J Transplant 2011;11:101-10. [PubMed]
- Boillot O, Delafosse B, Mechet I, et al. Small-for-size partial liver graft in an adult recipient; a new transplant technique. Lancet 2002;359:406-7. [PubMed]
- Cauchy F, Schwarz L, Scatton O, et al. Laparoscopic liver resection for living donation: where do we stand? World J Gastroenterol 2014;20:15590-8. [PubMed]
- Scatton O, Katsanos G, Boillot O, et al. Pure laparoscopic left lateral sectionectomy in living donors: from innovation to development in France. Ann Surg 2015;261:506-12. [PubMed]
- Boillot O, Sagnard P, Guillaud O, et al. Adult left liver transplantation from split livers and living donors: a 14-year single-center experience. Clin Transplant 2013;27:571-81. [PubMed]
- Spanish Society of Liver Transplantation. Spanish National Transplant Registry. Sociedad Espanola de Transplante Hepatico (SETH); Available online: www.sethepatico.org
- Sánchez-Cabús S, Abraldes JG, Taurá P, et al. Lack of correlation between preoperative and intraoperative liver hemodynamics: a descriptive analysis. Transplantation 2014;97:116-21. [PubMed]
- Sánchez-Cabús S, Fondevila C, Calatayud D, et al. Importance of the temporary portocaval shunt during adult living donor liver transplantation. Liver Transpl 2013;19:174-83. [PubMed]
- García-Valdecasas JC, Fuster J, Charco R, et al. Changes in portal vein flow after adult living-donor liver transplantation: does it influence postoperative liver function? Liver Transpl 2003;9:564-9. [PubMed]
- Hessheimer AJ, Escobar B, Munoz J, et al. Somatostatin therapy protects porcine livers in small-for-size liver transplantation. Am J Transplant 2014;14:1806-16. [PubMed]
- Garcia-Retortillo M, Forns X, Llovet JM, et al. Hepatitis C recurrence is more severe after living donor compared to cadaveric liver transplantation. Hepatology 2004;40:699-707. [PubMed]
- Ayuso JR, Ayuso C, Bombuy E, et al. Preoperative evaluation of biliary anatomy in adult live liver donors with volumetric mangafodipir trisodium enhanced magnetic resonance cholangiography. Liver Transpl 2004;10:1391-7. [PubMed]
- Herrero JI, Pardo F, Rotellar F, et al. Applicability of living donor liver transplantation in a program of adult liver transplantation. Transplant Proc 2011;43:690-1. [PubMed]
- Herrero JI, Rotellar F, Benito A, et al. Is liver biopsy necessary in the evaluation of a living donor for liver transplantation? Transplant Proc 2014;46:3082-3. [PubMed]
- Rotellar F, Pardo F, Benito A, et al. Totally laparoscopic right-lobe hepatectomy for adult living donor liver transplantation: useful strategies to enhance safety. Am J Transplant 2013;13:3269-73. [PubMed]
- Johnson RJ, Bradbury LL, Martin K, et al. Organ donation and transplantation in the UK-the last decade: a report from the UK national transplant registry. Transplantation 2014;97 Suppl 1:S1-S27. [PubMed]
- Organ Donation and Transplantation. UK Transplant Registry. Available online: http://www.odt.nhs.uk/uk-transplant-registry/
- NHS Organ Donation. NHS Organ Donor Register. Available online: https://www.organdonation.nhs.uk/faq/organ-donor-register/
- Dattani N, Baker A, Quaglia A, et al. Clinical and histological outcomes following living-related liver transplantation in children. Clin Res Hepatol Gastroenterol 2014;38:164-71. [PubMed]
- Scandiatransplant. Nordic liver transplant registry. Available online: http://www.scandiatransplant.org/data
- Olausson M, Mjornstedt L, Backman L, et al. Liver transplantation from a living donor is now possible. The first cases in Sweden completed with good results. Lakartidningen 1999;96:452-5. [PubMed]
- Fischler B, Johansson M, Gjertsen H, et al. Living-related liver transplantation in children--a single center evaluation of the outcome of donor candidates and recipients. Pediatr Transplant 2008;12:531-5. [PubMed]
- Olausson M, Mjornstedt L, Norden G, et al. Successful combined partial auxiliary liver and kidney transplantation in highly sensitized cross-match positive recipients. Am J Transplant 2007;7:130-6. [PubMed]
- Poltransplant. National Polish Transplant Registry. Available online: www.poltransplant.pl
- Paluszkiewicz R, Zieniewicz K, Kalinowski P, et al. Liver regeneration in 120 consecutive living-related liver donors. Transplant Proc 2009;41:2981-4. [PubMed]
- Mentha G, Morel P, Majno P, et al. Start of an adult living donor liver transplantation program in Switzerland. Schweiz Med Wochenschr 2000;130:1199-205. [PubMed]
- The EULOD project Living Organ Donation in Europe. Results and Recommendations. Available online: http://www.eulod.org/Files/Eulod/Content_Files/AKXVsAmbagtsheerWeimar%20EULOD_complete.pdf
- The EULID Project. Euro Living Donor Registry. Available online: www.eulivingdonor.eu/eulid
- The ELIPSY Project. Living donor psychosocial follow-up strategy and tools. Available online: www.eulivingdonor.eu/elipsy