Minimally invasive management of intrahepatic type II gallbladder perforation: a case report
Case presentation
A 70-year-old woman was addressed to the emergency department with a right upper quadrant abdominal pain. The patient reported an increasing abdominal pain over the last 10 days, associated with nausea and vomiting. She was admitted to hospital 3 months earlier for acute pancreatitis. She had a Balthazar grade E pancreatitis secondary to gallstone disease with two peripancreatic collections. Her previous admission was also complicated by a septic shock, which required a short stay in the intensive care unit and treatment with antibiotics. Her septicemia was caused by a central venous catheter infection. During her hospital stay, a CT-scan demonstrated the presence of gallbladder (GB) and common bile duct (CBD) stones. The patient underwent an endoscopic retrograde cholangiopancreatography (ERCP) with sphincterotomy, which led to the successful treatment of CBD stones. Three months after discharge, she underwent a CT-scan, which showed a regression of her pancreatitis (Balthazar grade D) with size reduction of the intra-abdominal collection. One month later, she came to the emergency department with abdominal pain, fever, and nausea. Blood tests showed an inflammatory syndrome with augmented C reactive protein (CRP) (300 mg/L) (normal values: <4 mg/L), hyperleukocytosis (18,000 g/L) (normal values: 4,000-10,500 g/L), and normal liver function tests.
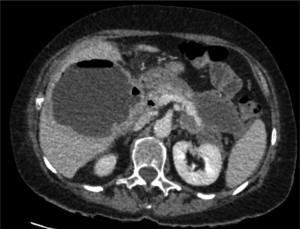
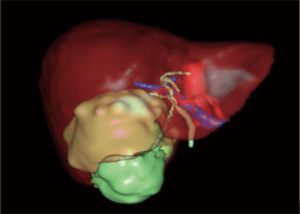
A CT-scan (Figure 1) and a 3D reconstruction (Figure 2) showed a large intrahepatic abscess associated with signs of a perforated GB secondary to acute cholecystitis. The peripancreatic collection was stable in size.
The patient was admitted to the hepatobiliary department, and antibiotherapy was initiated (ceftriaxone and metronidazole). A few hours after admission, the interventional radiologist decided to perform a percutaneous drainage (using a 16 French catheter), which successfully cleared the collection. Forty-eight hours later, culture results demonstrated the presence of two germs (Enterococcus avium and Enterococcus faecalis). Antibiotic therapy was further modified following the administration of vancomycin and cefepime. Abdominal pain decreased significantly 24 hours after drainage. Blood tests also showed that CRP levels decreased to 172.2 mg/L (36 hours after drainage) with a white blood cell count slightly elevated (15,000 g/L).
Six days after admission, CRP levels dropped down to 79.3 mg/L, and the patient was clinically less bothered by her abdominal pain and felt much less nauseous.
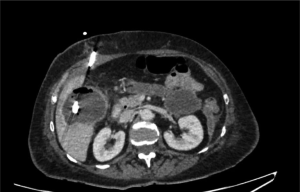
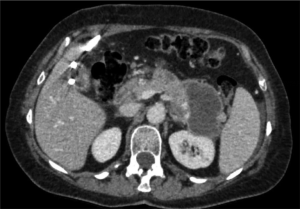
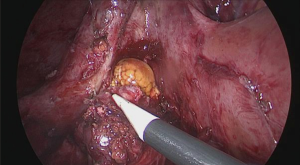
A control CT-scan (Figure 3) showed a reduction of the abscess and the adequate position of the drain. Ten days after hospital stay, the patient was discharged with her clamped drain in place and started to take amoxicillin and intravenous clavulanic acid. After the completion of the 21 days of antibiotics, a CT-scan (Figure 4) was performed. It showed the complete resolution of the hepatic abscess, which allowed for drain removal. Laparoscopic cholecystectomy was performed 3 months later (Figure 5) and the patient’s postoperative course was uneventful.
Discussion
Improvements in surgical and medical therapy have significantly reduced the risk of gallbladder perforation (GBP) (1,2). Historical study cohorts show that the rate of GBP secondary to acute cholecystitis was 10-15%, and currently decreased to a 0.8-3.2% (2). Additionally, different studies reported different mortality rates. Glene and Moore reported a 42% mortality rate whereas more recent studies reported a 12-16% mortality rate (3). In addition, an intrahepatic GBP with the development of an intrahepatic abscess is considered a rare medical entity, which had been reported a limited number of times in the medical literature (4). Liver abscess secondary to GBP is considered life-threatening with a 5.6% reported mortality rate (5). GBP was classified and described by Niemeier in three types (Table 1) (6).
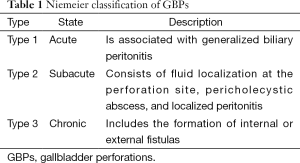
Full table
There are several mechanisms behind GBP. The most common mechanism is cystic duct obstruction, GB distension, altered vascularization, ischemia and necrosis followed by perforation (6). In the case of intrahepatic GBP with the development of a liver abscess, another mechanism is put forward. The other pathogenesis pathway starts with the infection of Rokitansky-Aschoff sinuses followed by necrosis, rupture and perforation (7). In addition, it is worthy to note that the fundus of the GB is the most common site of perforation secondary to its poor blood supply (6). Besides, it is suggested that people with a naturally partial or total intrahepatic GB are more likely to develop an intrahepatic abscess in case of GBP (7).
GBP can present differently depending on whether the GB perforated the abdominal cavity or other adjacent organs (5). A number of signs and symptoms can be present in GBP, including generalized abdominal pain or right upper quadrant pain, fever, right upper quadrant mass, or jaundice (4,6). In addition, patients can also present with septic shock and generalized peritonitis (6). GBP can be caused or associated with factors described in Table 1 (4,6,8). In addition, perturbation of liver function tests, with increased liver enzymes could be associated with this clinical situation (4). Usually, GBP can occur as early as 24 hours after the onset of acute cholecystitis or after a few days extending into weeks (9).
Ultrasound remains the preliminary investigation for biliary pathologies (10). Obesity, lack of patient’s cooperation, and meteorism can reduce the accuracy of sonography (11). Distended GB, ticked edematous GB pericholecystic fluid, and bulging of the GB are all considered non-specific signs of GBP (7). The “hole sign”, which is a defect in the GB wall is considered to be the only reliable sonographic sign of GBP (7). Evidence shows that most clinical series found that sonography is not as successful in detecting GBP as compared to CT-scan (10). CT-scan is considered the gold standard for the diagnosis of complicated cholecystitis (11). Signs of GBP on CT-scan include a focal defect in the GB wall, an interpretation of the GB wall or a gallstone outside the GB (9). CT-scan imaging can detect the interruption of the GB wall and the existence of extraluminal gallstones better than sonography (12).
As reported in the literature, the treatment of pyogenic hepatic abscesses includes antibiotics alone or antibiotics associated with a surgical therapy or a radiological intervention (13). In the modern era, the surgical management is becoming increasingly reserved to manage complications of failed radiological procedures (13). Successful management with antibiotics and percutaneous radiological drainage were shown in the study by Wong et al. which showed that 80 patients were all successfully treated without any surgical intervention (14). Wong treated hepatic abscesses of less than 5 cm with an antibiotic therapy and those above 5 cm with antibiotherapy and percutaneous drainage (14). Ferraioli et al. reviewed 148 patients aged over 21 and concluded that percutaneous drainage should always be undertaken before surgery (15,16). Ferraioli et al. demonstrated that percutaneous treatment was successful and none of the 104 patients who underwent this kind of therapy required a surgical procedure (15,16). In addition, Ferraioli et al. showed a significantly shorter hospital stay, a less expensive procedure with less morbidity in the 104 patients treated percutaneously as compared to patients treated surgically.
A large monocentric study identified factors, which can predict treatment failure, as described in Table 2 (17).

Full table
Conclusions
Intrahepatic GBPs represent a rare complication of acute and chronic GB disease, which requires a rapid treatment in order to reduce the pathology-related high morbidity and mortality. Considering the progress of interventional radiology, a rapid ultrasonography (US) or CT-scanning could facilitate the diagnosis. A percutaneous drainage and antibiotherapy could well treat the hepatic abscess followed by a distant cholecystectomy. Both procedures can be performed easily in most hospitals, hence avoiding more invasive procedures.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the editor-in-chief of this journal.
References
- Kochar K, Vallance K, Mathew G, et al. Intrahepatic perforation of the gall bladder presenting as liver abscess: case report, review of literature and Niemeier's classification. Eur J Gastroenterol Hepatol 2008;20:240-4. [PubMed]
- Göbel T, Kubitz R, Blondin D, et al. Intrahepatic type II gall bladder perforation by a gall stone in a CAPD patient. Eur J Med Res 2011;16:213-6. [PubMed]
- Ausania F, Guzman Suarez S, et al. Gallbladder perforation: morbidity, mortality and preoperative risk prediction. Surg Endosc 2015;29:955-60. [PubMed]
- Singh K, Singh A, Vidyarthi SH, et al. Spontaneous Intrahepatic Type II Gallbladder Perforation: A Rare Cause of Liver Abscess - Case Report. J Clin Diagn Res 2013;7:2012-4. [PubMed]
- Donati M, Biondi A, Basile F, et al. An atypical presentation of intrahepatic perforated cholecystitis: a modern indication to open cholecystectomy. Report of a case. BMC Surg 2014;14:6. [PubMed]
- Niemeier OW. Acute Free Perforation of the Gall-Bladder. Ann Surg 1934;99:922-4. [PubMed]
- Sood BP, Kalra N, Gupta S, et al. Role of sonography in the diagnosis of gallbladder perforation. J Clin Ultrasound 2002;30:270-4. [PubMed]
- Chowksey SR, Baghel H, Sharma P, et al. Diagnosis of Gallbladder Perforation-a Puzzle! Indian J Surg 2014;76:247-50. [PubMed]
- Bennett GL, Balthazar EJ. Ultrasound and CT evaluation of emergent gallbladder pathology. Radiol Clin North Am 2003;41:1203-16. [PubMed]
- Hutchings S, Punekar S, Wolstenhulme S. Gallbladder perforation: the hole sign. Ultrasound 2013;21:144-7.
- Tonolini M, Ravelli A, Villa C, et al. Urgent MRI with MR cholangiopancreatography (MRCP) of acute cholecystitis and related complications: diagnostic role and spectrum of imaging findings. Emerg Radiol 2012;19:341-8. [PubMed]
- Seyal AR, Parekh K, Gonzalez-Guindalini FD, et al. Cross-sectional imaging of perforated gallbladder. Abdom Imaging 2014;39:853-74. [PubMed]
- Pang TC, Fung T, Samra J, et al. Pyogenic liver abscess: an audit of 10 years' experience. World J Gastroenterol 2011;17:1622-30. [PubMed]
- Wong WM, Wong BC, Hui CK, et al. Pyogenic liver abscess: retrospective analysis of 80 cases over a 10-year period. J Gastroenterol Hepatol 2002;17:1001-7. [PubMed]
- Ferraioli G, Garlaschelli A, Zanaboni D, et al. Percutaneous and surgical treatment of pyogenic liver abscesses: observation over a 21-year period in 148 patients. Dig Liver Dis 2008;40:690-6. [PubMed]
- Tan YM, Chung AY, Chow PK, et al. An appraisal of surgical and percutaneous drainage for pyogenic liver abscesses larger than 5 cm. Ann Surg 2005;241:485-90. [PubMed]
- Lo JZ, Leow JJ, Ng PL, et al. Predictors of therapy failure in a series of 741 adult pyogenic liver abscesses. J Hepatobiliary Pancreat Sci 2015;22:156-65. [PubMed]