
Heterogenous responses to nivolumab in a single metastatic nodule in hepatocellular carcinoma: role of salvage surgery
A recent report demonstrated immune heterogeneity of hepatocellular carcinoma (HCC) (1). The objective response rates of sorafenib-treated global and Asian patients by nivolumab, the monoclonal antibody to programmed cell death protein-1, were only 14% and 15%, respectively (2,3). However, there is a lack of reports demonstrating the heterogenous responses within single tumor to nivolumab. Here, we show a pathologically confirmed, heterogenous response to nivolumab within a single metastatic tumor from HCC. In these circumstances, surgery can be considered as an option to cure the disease.
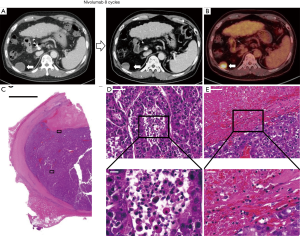
A 61-year-old Korean man with a history of hepatitis B virus-related cirrhosis and HCC who was previously treated with hepatic arterial infusion chemotherapy and transarterial radioembolization (TARE) with Yittrium-90 (Y-90) developed numerous multiple lung and single nodular peritoneal metastases. The intrahepatic lesion showed a complete radiological response by computed tomography (CT) after TARE. After TARE with Y-90, his alpha-fetoprotein (AFP) and protein induced by vitamin K absence (PIVKA)-II levels were 13,985 ng/mL and 9,027 mAU/mL, respectively. The patient was initially treated with 800 mg of sorafenib daily. After 8 weeks of sorafenib treatment, he was diagnosed with progressive disease because of increased size of a peritoneal metastasis (4.0 cm) and aggravation of multiple lung metastases. He was then started on nivolumab (3 mg/kg every two weeks). After 8 cycles of nivolumab, his lung metastasis disappeared, although the remnant peritoneal metastasis (2.8 cm) had an increasing arterial-enhancing portion with non-enhancing necrotic portion (Figure 1A). 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET) showed an FDG-avid peritoneal nodule with no FDG uptake in the anterior one-third portion (Figure 1B). His AFP and PIVKA-II levels were markedly decreased to 68.3 ng/mL and 71 mAU/mL, respectively. The surgical team decided to perform metastasectomy to cure the disease. The patient underwent laparoscopic mass excision for the peritoneal metastatic nodule. The pathologic specimen showed metastatic HCC with partial necrosis (Figure 1C). The FDG-avid portion of peritoneal nodule consisted of viable tumor cells with various types of tumor-infiltrating immune cells (Figure 1D). In the necrotic portion, immune cells were also infiltrated, particularly near the border between the viable tumor and necrosis (Figure 1E). Two months after surgery, there was no evidence of recurrence in the abdomen or lung CT scans.
Discussion
Intratumoral heterogeneous responses to immune checkpoint inhibitors can be explained by innate or acquired resistance of tumor cells (4). In our case, the different tumor responses between lung and peritoneal metastasis and the different responses to nivolumab treatment within the single peritoneal metastatic nodule may reflect the immune heterogeneity of HCC. In our case, surgery was performed and the patient became free of the disease. The pathology specimen of the peritoneal metastasis showed that tumor infiltrating immune cells induced necrosis in part but did not in another portion. Interestingly, various immune cells were still infiltrated between viable tumor cells, suggesting that these cells may be resistant to “functionally active” anti-tumor T cells. Surgical resection, not more rounds of nivolumab treatment, resulted in the complete response. Immune heterogeneity should be considered when treating HCC with immune checkpoint inhibitors.
The ratio of T-cell invigoration to tumor burden ratio has been reported to be associated with the response in melanoma patients treated with pembrolizumab (5). In our case, the maximal diameter of metastatic nodules in the lung, which was successfully treated with nivolumab, was less than 1.0 cm. On the other hand, the diameter of peritoneal metastasis, which contained viable tumor cells resistant to nivolumab, was 4.0 cm. Immune heterogeneity can be applied to the larger tumors that may contain more resistant clones to immune checkpoint inhibitors.
In summary, our case demonstrates that single tumor nodule of HCC can also show heterogenous responses to nivolumab, and surgical resection of that nodule can be chosen as the optimal salvage treatment.
Acknowledgments
Funding: This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (S.P.S) (NRF-2019R1I1A1A01059642).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Zhang Q, Lou Y, Yang J, et al. Integrated multiomic analysis reveals comprehensive tumour heterogeneity and novel immunophenotypic classification in hepatocellular carcinomas. Gut 2019. [Epub ahead of print]. [Crossref] [PubMed]
- Yau T, Hsu C, Kim TY, et al. Nivolumab in advanced hepatocellular carcinoma: Sorafenib-experienced Asian cohort analysis. J Hepatol 2019;71:543-52. [Crossref] [PubMed]
- El-Khoueiry AB, Sangro B, Yau T, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017;389:2492-502. [Crossref] [PubMed]
- Jenkins RW, Barbie DA, Flaherty KT. Mechanisms of resistance to immune checkpoint inhibitors. Br J Cancer 2018;118:9-16. [Crossref] [PubMed]
- Huang AC, Postow MA, Orlowski RJ, et al. T-cell invigoration to tumour burden ratio associated with anti-PD-1 response. Nature 2017;545:60-5. [Crossref] [PubMed]


