Management of unresectable intrahepatic cholangiocarcinoma: how do we decide among the various liver-directed treatments?
Introduction
The growing number of treatment options for intrahepatic cholangiocarcinoma
Cholangiocarcinoma is a disease with high mortality rates largely owing to its infiltrative nature, propensity for advanced disease presentation, and resistance to chemotherapy. Indeed, only 12% of all patients have localized disease at presentation, and of these patients with localized disease, less than 40% undergo cancer-directed surgery (1). This leaves a large proportion of patients with liver-confined, localized disease with several options for liver-directed therapies, usually after undergoing initial systemic chemotherapy. These options include transarterial chemoembolization (TACE), hepatic arterial infusion (HAI), percutaneous ablation [e.g., radiofrequency ablation (RFA)], external beam radiation therapy (EBRT), and radioembolization (RE). As yet, there has been no randomized clinical trial to compare these various treatment options for intrahepatic cholangiocarcinoma. Here, we review the most updated results of these treatment modalities for patients with unresectable disease and discuss the potential roles for each in the era of personalized medicine.
The importance of local control
A notable feature of unresectable intrahepatic cholangiocarcinoma is that the majority of patients die of tumor-related liver failure (2); while extrahepatic distant metastasis is prevalent, it is not as frequently the cause of death as the intrahepatic disease. Inadequate control of the primary tumor and satellite lesions can lead to parenchymal loss and liver failure due to vascular compromise (portal venous or hepatic vein obstruction) or biliary obstruction (potentially leading to sepsis) (3). Our review of patients with unresectable intrahepatic cholangiocarcinoma who received radiotherapy at MD Anderson Cancer Center from 2002 to 2014 revealed that a bioequivalent dose less than 80.5 Gy BED led to poor tumor control. Tumor progression led to tumor-related liver failure in 89% of the patients whose cause of death could be determined. In these patients, half of them died from tumor-related biliary obstruction and the rest died of vascular compromise or a combination of both. These results strongly emphasize the need to develop effective liver-directed therapies to achieve local control in patients with unresectable disease. We have seen similar results in a larger cohort of patients with liver-confined intrahepatic cholangiocarcinoma treated at our institution. In this study, we noted improvements over time with resection and radiotherapy, likely owing to technical improvements in these treatments. On multivariable analysis, receipt of local therapy (with resection or definitive radiotherapy) was the sole predictor of death without liver failure (3).
Overview of liver-directed therapy options
Surgical resection represents the gold standard for liver-directed therapy, but is an option for only a small proportion of patients. For the small subset of patients with small, inoperable, and peripheral tumors, percutaneous image-guided ablative options like RFA and microwave ablation are convenient and cost effective (4,5). These achieve high rates of local tumor control and prolong survival for inoperable patients (6). Additional options include arterial embolization with yttrium-90 (90Y) (7-9) and transarterial chemoembolization (10,11). Although cholangiocarcinoma is generally regarded as a hypovascular tumor in which transarterial delivery may be suboptimal, these options have been studied for a wide variety of patients and in various clinical scenarios. In limited series, TACE and 90Y may be useful options in patients for whom surgical resection may be considered if an adequate response is achieved. These therapies are also useful as a bridging therapy for transplant, or as definitive therapy in properly selected patients.
Radiation therapy is another treatment option for patients with intrahepatic cholangiocarcinoma and can be delivered in any region of the liver effectively. The radiation can be administered either with brachytherapy techniques or external beam radiation. Some specialized centers have practiced interstitial brachytherapy for liver tumors for over a decade (12-15), showing that one year local control rates above 90% can be achieved with this approach. However, Longer-term reports of interstitial brachytherapy are not available.
Our previous work has described how external beam radiation may be a reliable and effective option for centrally located tumors near hilar structures and for tumors near or involving the main portal vein or inferior vena cava (2,16). Tumors in these locations are often not ideal for resection or percutaneous ablation but are at a high risk of causing liver failure related to vascular or biliary compromise. We will discuss these points in more detail in sections dedicated to each liver-directed therapy.
Endpoints for liver-directed therapies
In the absence of randomized data, we are left with historical comparisons of various liver-directed therapies, making it difficult to truly ascertain which may be best in a given scenario. Patient selection and baseline characteristics are important factors to consider in performing retrospective or post-hoc analyses. Another critical consideration in these comparisons is the endpoint measurement for each study. Given that intrahepatic cholangiocarcinoma can directly cause liver failure in patients if the primary disease or satellite lesions are not controlled, local control of the primary tumor and intrahepatic control are arguably the most relevant endpoint measurements for liver-directed therapies.
While many experts may acknowledge that these endpoints are important, there is still considerable heterogeneity in the quality of endpoint measurements in the literature pertinent to intrahepatic cholangiocarcinoma. For example, the traditional endpoint for external beam radiation therapy is local control (i.e., did the tumor progress locally or not after radiation therapy?). We have assessed this based on expert radiographic assessment including assessment of tumor growth by RECIST criteria but also consideration of whether there are clinical signs of local progression (2). Local control is an actuarial measurement, allowing investigators to know the expected duration of effect.
For other liver-directed therapies such as TACE, RFA, and radioembolization, it is common to report outcomes in terms of various radiographic response criteria that are available, including Response Evaluation Criteria in Solid Tumors (RECIST) and European Association for the Study of Liver (EASL) criteria (Figure 1). These response criteria were mostly developed and applied to HCC (17-19). Retrospective evaluation of modified RECIST (mRECIST) have been performed with intra-arterial therapies for intrahepatic cholangiocarcinoma (20,21). These liver-directed therapies are also often reported in terms of overall survival. The endpoint measurements of radiographic response and overall survival would be most useful if they have a clear relevance to the effectiveness of the liver-directed therapy (i.e., is there a direct relationship between therapeutic efficacy with radiographic response and does this translate into prolonged overall survival?). Further, the response criteria of RECIST, mRECIST, and EASL still require prospective validation in the specific contexts of TACE, RFA, or radioembolization for intrahepatic cholangiocarcinoma. Currently, the largest reports of outcomes after these liver-directed therapies report response in more than one measurement or in different methods from each other.
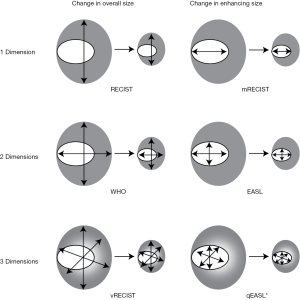
RECIST criteria, which are still reported in modern series as primary endpoint measurements, are particularly fraught with problems for liver tumors. In 2001, the European Association for the Study of the Liver conference (22) concluded that RECIST criteria were not optimal for measuring response to liver-directed therapies. For example, the RECIST criteria failed to identify all of the complete responses in two prospective cohorts, meaning that the efficacy of treatment would not have been appreciated (23). The EASL criteria were developed as a result of these important studies, but these criteria face challenges for cholangiocarcinoma, especially when the tumors are hypovascular. In one study, 28% of patients with intrahepatic cholangiocarcinoma treated with TACE could not be assessed for response by EASL or modified RECIST due to enhancement patterns (24).
The key problem with response criteria as surrogate endpoints is that they fail to provide an actuarial measurement. Although a patient may achieve a partial response by RECIST or EASL criteria, the important question is how much time the tumor is controlled, and if such control leads to extended overall survival. Going forward, as the clinical evaluations of ablative external beam radiotherapy and other liver-directed therapies proceed, radiographic response criteria should be uniformly employed and subjected to rigorous standards (25) to ensure that the measurements are reliable, reproducible, and strongly correlated with clinical outcomes. Until these radiographic criteria meet the proper standards as validated surrogate endpoints for each liver-directed treatment, we encourage investigators to systematically follow their patients who receive liver-directed therapies and measure local control and intrahepatic control as endpoints.
Liver-directed therapies
General considerations in personalizing liver-directed therapy
The critical point to consider in personalizing therapy for a patient is the therapeutic ratio: what is the expected benefit compared to the risks? Factors that influence the answer to this question include the patient’s general medical condition and comorbidities (especially in regards to underlying liver function), the patient’s treatment history, the size of the tumor, the vascularity of the tumor, and the location of the tumor (particularly in relation to bile ducts, blood vessels, bowel, diaphragm, chest wall, and gall bladder). Finally, the clinical evidence should also factor into decision-making. There is substantial heterogeneity in the use of different liver-directed therapies and generally low-level evidence to support their use in any given situation. Since randomized data that directly compare different liver-directed therapies are not available, we focus on each liver-directed treatment to help create a general rubric for personalized therapy.
Transarterial chemoembolization (TACE)
Among liver-direct therapies, TACE has been used with a high level of success with hepatocellular carcinoma (HCC) for various indications. Investigations of TACE for intrahepatic cholangiocarcinoma has been of great interest for the past decade, but few prospective studies have been performed. Most studies are retrospective, single institution reports. One prospective study of 26 patients (23 with intrahepatic cholangiocarcinoma and 3 with gallbladder cancer) using chemoembolization with irinotecan-eluting beads was conducted by Kuhlmann et al. showing a median progression free survival (PFS) of 3.9 months and overall survival (OS) of 11.7 months. By comparison, a historical cohort of 10 patients who received TACE with mitomycin C had PFS of 1.8 months and OS of 5.7 months, and another historical cohort of 31 patients who received gemcitabine and oxaliplatin had PFS of 6.2 months and OS of 11.0 months (11).
Retrospective studies of TACE have reported a range of survival times for limited numbers of patients, including an OS of 12.3 months with cisplatin (n=50) (26), 13 months with doxorubicin microspheres (n=11) (27), 21.1 months with mitomycin C (n=15) (28), 23 months with cisplatin, doxorubicin, and mitomycin C (n=17) (29), 30 months (n=9) (30), 15 months with cisplatin, doxorubicin, and mitomycin C (n=62) (31), and 13 months with variable regimens including mitomycin-C, gemcitabine, and cisplatin (n=115) (32). These studies examined a diverse group of patients in terms of patient and tumor characteristics, likely accounting for the wide range of outcomes that were reported. The selection of patients for TACE, in general, may depend on patient performance status, baseline liver function, presence of constitutional symptoms (weight loss, malaise, loss of appetite), vascularity of the tumor, absence of portal vein tumor thrombus, absence of extrahepatic disease, and size of the tumor (33).
Hepatic arterial infusion (HAI)
HAI has been studied mostly in colorectal liver metastases with more limited data being available for intrahepatic cholangiocarcinoma (34-37). The reported median survival times with HAI for intrahepatic cholangiocarcinoma range from 12.5 (35) to 31.1 months (37). Two modern studies were performed by Kemeny and associates. The first included patients with unresectable intrahepatic cholangiocarcinoma or hepatocellular carcinoma who received HAI with floxuridine (FUDR)/dexamethasone (median survival of 29.5 months, n=26 intrahepatic cholangiocarcinoma and 8 HCC) (36), and the second involved similar patients who received HAI with intravenous bevacizumab (median survival of 31.1 months, n=18 intrahepatic cholangiocarcinoma and 4 HCC) (37). The trial with bevacizumab was stopped early due to biliary toxicity requiring stents in 13.6% of patients. Generally, selection of patients for this procedure requires good baseline liver function, blood counts, performance status, as most other liver-directed therapies. The procedure is generally not done for patients with portal hypertension or portal inflow occlusion.
Percutaneous ablation
RFA and microwave ablation achieve tumor kill largely through thermal effects on the cancer cells. These treatments have been an effective modality for small liver tumors, including hepatocellular carcinoma and liver metastases. The use of RFA for intrahepatic cholangiocarcinoma has been studied only in retrospective series. One of the largest series reviewed the outcomes of 13 patients with 17 intrahepatic cholangiocarcinoma tumors treated with RFA at a single institution. The local progression free survival was 32.2 months. Most of the tumors were small, with 10 of the treated tumors having a diameter less than 3 cm, five of the tumors measuring between 3 and 5 cm, and two measuring larger than 5 cm. Treatment failure occurred in the two largest tumors. The median overall survival of these patients was 38.5 months, the 3-year survival rate was 51%, and the 5-year survival rate was 15% (38). A retrospective study of sonography-guided microwave ablation included 15 patients with a mean tumor size of 3.2 cm (range, 1.3–9.9 cm) (39). The 24 month survival rate was 60% in this study. Two patients developed liver abscess, and one had needle seeding. Another retrospective study included 18 patients with 25 intrahepatic cholangiocarcinoma tumors who underwent either RFA or microwave ablation. The 36 month survival rate was 30.3% (40).
A recent systematic review on the use of RFA for the treatment of unresectable intrahepatic cholangiocarcinoma identified seven observational studies comprising 84 patients (6). Pooled 1-, 3-, and 5-year survival rates were 82% (95% CI, 72–90%), 47% (95% CI, 28–65%), and 24% % (95% CI, 11–40%), respectively. Major complications occurred in 5 patients, including one death from liver abscess and subsequent sepsis.
A new ablation technique called irreversible electroporation (IRE) can also achieve high rates of local tumor control, including for lesions near large vessels and bile ducts. This is due to a different mechanism of cell kill that does not rely on thermal damage. IRE uses electrical fields to cause permanent nanopores in the cell membranes of cells that leads to apoptosis. It does not seem to cause damage to adjacent bile ducts or vessels (41). The use of IRE with cholangiocarcinoma has been limited, and large series remain to be seen.
The complications with percutaneous treatments are minimal in well-selected patients. Caution with thermal treatments (RFA and microwave ablation) must be taken to avoid tumors near a segmental bile duct, however. This can result in bile leak, which can be life-threatening. It is also important to understand that nearby vessels may act as a heat sink for RFA and microwave ablation, limiting efficacy.
The general indications to use these percutaneous RFA and microwave ablation are for patients with tumors less than 4 to 5 cm that are not near a segmental bile duct, liver surface, or major vessel. IRE does not seem to have the same limitations as RFA and microwave ablation in regards to proximity to bile ducts or vessels, but is limited by tumor size. The literature indicates that attempted percutaneous ablation of larger HCC tumors results in a higher failure rate (42).
Radioembolization (RE)
Among the liver-directed therapies, yttrium-90 (90Y) microspheres have received the most attention. In a group of 60 patients who were prospectively followed after RE, the median survival was 15.6 months for patients with peripheral tumors and 6.1 months for those with an infiltrative morphology (43). For 5 patients in this study, the disease was converted to resectable status, and an R0 resection was achieved. A pooled analysis of 12 studies found a median weighted overall survival of 15.5 months after 90Y treatment (9). Partial response was seen in 28% of patients, and 54% of patients achieve stable disease at 3 months.
One limitation of RE is the delivery of sufficient microspheres to achieve complete ablation of the tumor. This therapy is also challenged by the lack of a validated method to measure the dose of radiation that is delivered to the tumor. For example, Bremsstrahlung imaging methods have been developed to address the dosimetry (44), but remains to be fully utilized in clinical practice. Other methods have been developed also, and each has uncertainties (45). Currently, the method of prescription for 90Y is with an amount of radiation activity. This method fails to determine how much radiation dose is given to the tumor in an individual patient. In external radiation treatments, evidence suggests a dose-response relationship (2), and one can expect that with internal radiation treatment with 90Y for intrahepatic cholangiocarcinoma a similar relationship exists. This relationship is likely dependent on the arterial supply of the tumor. Indeed, for hepatocellular carcinoma, which is typically dependent on an arterial supply, dose-response relationships have been reported (46,47). Development of a method to measure the dose delivered to the tumor a priori would help in the proper selection of patients for this treatment modality.
Brachytherapy
Small single institutional series have been conducted to show that primary and metastatic liver cancers can be safely treated with interstitial brachytherapy. One report for HCC included large liver tumors (mean size of 7.1 cm), and achieved a local control rate of 93% at one year (14). Reports of this technique with metastatic lesions have also been published (12,13,15). To date, there are no dedicated studies describing the efficacy of this technique specifically for intrahepatic cholangiocarcinoma. Among the liver-directed therapies, this form of treatment is perhaps one of the most technically challenging, as it not only requires some degree of hands-on skill, but also appropriate facilities and technical support to perform.
External beam radiation
Decades of clinical research and technological advancement has shown that ablation of tumors in organs with parallel functional units, such as lung and liver, can be done safely. A series of studies in the 1980s, 1990s and 2000s developed the techniques, dose constraints, and clinical indications for delivery of stereotactic body radiation therapy (SBRT, also called stereotactic ablative body radiotherapy or SABR). Traditional SBRT achieves excellent results for small liver tumors (<5 cm). However, the vast majority of physicians interpret SBRT as meaning doses of radiation (4 to 20 Gy) that may not be ablative, but are delivered within about 1 week (i.e., in 3 to 6 fractions). Adherence to this approach has limited the effectiveness of SBRT for large liver tumors (>7 cm) owing to the need to reduce doses to meet organ constraints. With chemotherapy alone, the prognosis for patients who present with large liver tumors is poor, with median survival times of 12 months or less, and most such patients die from tumor-related liver failure. We have presented a comprehensive solution that achieves stereotactic ablative body radiation (SABR) doses for patients with large liver tumors by using a combination of classical, modern, and novel concepts of radiotherapy: fractionation, dose painting, motion management, image guidance, and simultaneous integrated protection (16).
These concepts were partly pioneered with proton therapy in Japan for HCC. Protons have allowed large tumors to be treated to higher doses per fraction. The reports of experiences with hypofractionated regimens (16–25 fractions) that go to ablative doses (BED ~100 Gy) for large tumors are similar to those after surgical resection, with 5-year local tumor control rates of 90% and overall survival (OS) rates of 50% among some patients (48-50). These studies generally selected patients with relatively small, isolated tumors with well-compensated cirrhosis.
Building on these successes in Japan, we began patterning our treatment of patients with liver tumors using a fractionated approach to high doses in 2007. A retrospective dose response analysis of patients given definitive radiation therapy for inoperable intrahepatic cholangiocarcinoma in 2002–2014 at MD Anderson was recently published (2), and it identified 79 consecutive patients, most of whom had large tumors [median diameter 7.9 cm (range, 2.2–17 cm)]. Seventy patients (89%) had received systemic chemotherapy before radiation, which was given to doses of 35–100 Gy (median 58.05 Gy), for a median BED of 80.5 Gy (range, 43.75–180 Gy). At a median follow-up time of 33 months (range, 11–93 months), the median OS time after diagnosis was 30 months and the 3-year OS rate was 44%. Radiation dose was the single most important prognostic factor; higher doses correlated with improved local control and OS. The 3-year OS rate for those receiving BED >80.5 was 73% versus 38% for those receiving lower doses (P=0.017), and the 3-year local control rate was significantly higher (78%) after a BED >80.5 Gy than after lower doses (45%, P=0.04). As a continuous variable, BED also significantly influenced local control (P=0.0097) and OS (P=0.0045). No significant treatment-related toxicity was noted. These results suggest that a BED >80.5 Gy seems to be ablative for large intrahepatic cholangiocarcinomas, with long-term survival rates that compare favorably to resection. Achieving these results requires meticulous attention to motion management and image guidance using conformal radiation techniques such as intensity modulated radiation therapy and proton therapy.
A path forward
Choosing among the different liver-directed therapies
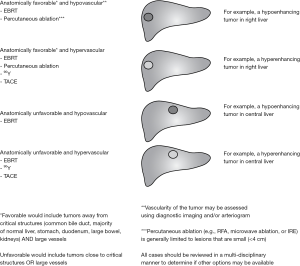
In the absence of randomized studies that directly compare the various options for liver-directed treatments, we propose a clinical algorithm that provides a path forward until such studies are available or until the weight of clinical evidence steers the field in the right direction (Figure 2). This algorithm takes clinical and tumor factors into consideration. Importantly, the algorithm points to multiple options that may be well suited for an individual, and the ultimate decision should involve multi-disciplinary discussion.
For example, in patients with small (<4 cm), peripherally located, enhancing tumors, percutaneous ablation is a reasonable option, since such tumors would be unlikely to be near major blood vessels and medium to large bile ducts. TACE, RE, and radiotherapy may all be reasonable considerations in larger (>4 cm) peripherally located, enhancing tumors. These tumors would be expected to have dependence on an arterial blood supply so that intra-arterial therapeutic delivery would be adequate, and would likely be away from critical organs that would be sensitive to radiation. Factors that may influence the final decision may include the patient’s co-morbid conditions and prior treatment history.
On the other end of the spectrum, patients with large, central, hypovascular tumors that are adjacent to major blood vessels would likely be best treated with external beam radiation. These tumors would unlikely respond to trans-arterial therapies, and RFA would have limited efficacy due to the size of the tumor, heat sink effect of the blood vessels, and risk of bile duct injury. For patients with small central tumors, percutaneous ablation with IRE is also a consideration, including for tumors adjacent to large vessels and bile ducts. Our approach of using fractionated radiation to ablative doses is generally well tolerated in these locations with no major complications (16).
Defining target volumes for liver-directed therapies
One technical aspect that may improve outcomes with all liver-directed therapies is the definition of the tumor volume. The delineation of tumor boundaries for intrahepatic cholangiocarcinoma may be a major limiting factor for all liver-directed treatment options because missing any part of the tumor with a cytotoxic or ablative therapy would be expected to lead to a local failure. Sensitive and specific diagnostic imaging tests to identify the boundaries of disease remain elusive for intrahepatic cholangiocarcinoma. To address this limitation, it will be critical to use radiological/pathological correlation studies to understand the true burden of disease for patients whose treatment can only be guided by radiological studies. It is also crucial to consider the patterns of spread for these tumors.
On microscopic examination, 90% of cholangiocarcinomas are adenocarcinomas with glandular structures mixed with fibrous stroma (51). These tumors preferentially spread between hepatic plates and track along the bile ducts (52). Perineural invasion is seen in about 81% of the patients (53). On imaging, intrahepatic cholangiocarcinoma appears as a hypodense mass with irregular rolled in margins. On contrast enhanced images, the masses may show irregular peripheral enhancement (54). Due to the abundant fibrous stroma, IHCC tumors typically show higher enhancement in the portal venous and delayed images (55). The development of advanced MRI and molecular imaging agents may help oncologists better define tumor volumes on imaging (55-57).
Quantitative response criteria
As previously discussed in Section I, the response criteria to liver-directed therapies represents a major limiting factor in the comparison of the various options. An objective and reproducible method that represents a surrogate to local control and overall survival is needed. Several solutions have emerged in recent years.
Geschwind and associates have published as series of papers that describe how tumor viability can be gauged based on how the degree of enhancement in the tumor changes with cytotoxic treatments (called quantitative EASL or qEASL) (58,59). In primary and secondary lesions of the liver, the measurement of normalized enhancement in the tumor serves as a quantitative readout of response to therapy that associates with overall survival. This group has described the application of criteria to stratify patients by converting 2D response criteria into 3D response criteria (58,59). Proper prospective validation of these quantitative approaches for each liver-directed treatment could help to address the lingering issue for oncology with regard to which liver-directed therapy is indicated in certain clinical scenarios.
Molecular oncology and combined liver-directed therapies
The biological heterogeneity of the disease is perhaps the most important driver of the variety of presentations and treatment outcomes for patients. Recent studies show that the disease is molecularly diverse with several subclasses of patients, including those with increased signaling in EGFR and HER2 pathways, as well as patients with actionable mutations in IDH1, FGFR2, and BRAF (60,61). These different types of cholangiocarcinoma have distinct prognoses, and the development of non-invasive or minimally invasive methods to characterize the disease are needed. This is relevant not only to the development of new and more efficacious systemic therapies for the disease, but also to the selection of liver-directed therapies or how they are delivered. First, some cholangiocarcinomas that may be associated with more indolent courses or liver-confined patterns of failure may be better suited to early aggressive liver-directed therapies, whereas tumors with a greater propensity for distant metastatic spread may not. Additionally, tumors with genetic susceptibility to DNA damage repair may benefit from radiotherapy and/or targeting DNA repair pathways. Similarly, a molecular understanding of the disease could aid the development of combinations of liver-directed and systemic therapies, concurrently or sequentially. There are currently no published randomized trials that compare local therapies against systemic therapies. The ongoing NRG-GI001 trial will attempt to investigate this question, as it randomizes patients to either systemic chemotherapy followed by hypofractionated radiotherapy or systemic chemotherapy alone.
Finally, innovative combinations of liver-directed therapies may be warranted. For example, TACE and external beam radiotherapy have been used with success in HCC. One may also conceive combining RE with external beam radiotherapy, such as in tumors that are near critical organs that can be damaged by radiotherapy but have limited arterial supply. Complete coverage of the tumor with ablative radiation doses may be achieved in such a situation with the combination of internal and external radiotherapy.
Summary
Progress is being made for patients with intrahepatic cholangiocarcinoma as multiple liver-directed therapies are being reported to achieve long-term survival. In our institution, we have seen a steady improvement in survival outcomes for patients with liver-confined disease, likely owing to improved surgical and radiotherapy techniques (3). However, further progress is severely hampered by the lack of a concerted effort to appropriately evaluate these therapies in terms of their indications and efficacy. In particular, prospective trials with rigorous selection criteria, response metrics, and actuarial outcome measurements of local control and overall survival are needed. Ideally, these studies would be randomized trials that directly compare liver-directed treatments, but this may be challenging for various reasons, including patient resistance to randomization, physician bias, and lack of expertise in particular centers. Innovative trial designs and testing of combinations of liver-directed therapies are also needed, especially in the context of molecular oncology.
Acknowledgements
Funding: This work was supported in part by Cancer Center Support (Core) Grant CA016672 from the National Cancer Institute to The University of Texas MD Anderson Cancer Center. Eugene Koay was supported in part by grants from the University of Texas MD Anderson Cancer Center including the Cancer Moonshots, from the MD Anderson Center for Advanced Biomedical Imaging with equipment support from GE Healthcare, and by a sponsored research agreement with Philips Healthcare.
Footnote
Conflicts of Interest: Eugene Koay received an honorarium from Philips Healthcare for speaking at a Philips-sponsored event during the annual Radiological Society of North America meeting in 2015. The other authors have no conflicts of interest to declare.
References
- Tan JC, Coburn NG, Baxter NN, et al. Surgical management of intrahepatic cholangiocarcinoma--a population-based study. Ann Surg Oncol 2008;15:600-8. [Crossref] [PubMed]
- Tao R, Krishnan S, Bhosale PR, et al. Ablative Radiotherapy Doses Lead to a Substantial Prolongation of Survival in Patients With Inoperable Intrahepatic Cholangiocarcinoma: A Retrospective Dose Response Analysis. J Clin Oncol 2016;34:219-26. [Crossref] [PubMed]
- Yamashita S, Koay EJ, Passot G, et al. Local therapy reduces the risk of liver failure and improves survival in patients with intrahepatic cholangiocarcinoma: A comprehensive analysis of 362 consecutive patients. Cancer 2017;123:1354-62. [PubMed]
- Kim JH, Won HJ, Shin YM, et al. Radiofrequency ablation for the treatment of primary intrahepatic cholangiocarcinoma. AJR Am J Roentgenol 2011;196:W205-9. [Crossref] [PubMed]
- Zgodzinski W, Espat NJ. Radiofrequency ablation for incidentally identified primary intrahepatic cholangiocarcinoma. World J Gastroenterol 2005;11:5239-40. [PubMed]
- Han K, Ko HK, Kim KW, et al. Radiofrequency ablation in the treatment of unresectable intrahepatic cholangiocarcinoma: systematic review and meta-analysis. J Vasc Interv Radiol 2015;26:943-8. [Crossref] [PubMed]
- Ibrahim SM, Mulcahy MF, Lewandowski RJ, et al. Treatment of unresectable cholangiocarcinoma using yttrium-90 microspheres: results from a pilot study. Cancer 2008;113:2119-28. [Crossref] [PubMed]
- Saxena A, Bester L, Chua TC, et al. Yttrium-90 radiotherapy for unresectable intrahepatic cholangiocarcinoma: a preliminary assessment of this novel treatment option. Ann Surg Oncol 2010;17:484-91. [Crossref] [PubMed]
- Al-Adra DP, Gill RS, Axford SJ, et al. Treatment of unresectable intrahepatic cholangiocarcinoma with yttrium-90 radioembolization: a systematic review and pooled analysis. Eur J Surg Oncol 2015;41:120-7. [Crossref] [PubMed]
- Kim JH, Yoon HK, Sung KB, et al. Transcatheter arterial chemoembolization or chemoinfusion for unresectable intrahepatic cholangiocarcinoma: clinical efficacy and factors influencing outcomes. Cancer 2008;113:1614-22. [Crossref] [PubMed]
- Kuhlmann JB, Euringer W, Spangenberg HC, et al. Treatment of unresectable cholangiocarcinoma: conventional transarterial chemoembolization compared with drug eluting bead-transarterial chemoembolization and systemic chemotherapy. Eur J Gastroenterol Hepatol 2012;24:437-43. [PubMed]
- Ricke J, Wust P, Wieners G, et al. Liver malignancies: CT-guided interstitial brachytherapy in patients with unfavorable lesions for thermal ablation. J Vasc Interv Radiol 2004;15:1279-86. [Crossref] [PubMed]
- Tselis N, Chatzikonstantinou G, Kolotas C, et al. Computed tomography-guided interstitial high dose rate brachytherapy for centrally located liver tumours: a single institution study. Eur Radiol 2013;23:2264-70. [Crossref] [PubMed]
- Collettini F, Schnapauff D, Poellinger A, et al. Hepatocellular carcinoma: computed-tomography-guided high-dose-rate brachytherapy (CT-HDRBT) ablation of large (5-7 cm) and very large (>7 cm) tumours. Eur Radiol 2012;22:1101-9. [Crossref] [PubMed]
- Collettini F, Singh A, Schnapauff D, et al. Computed-tomography-guided high-dose-rate brachytherapy (CT-HDRBT) ablation of metastases adjacent to the liver hilum. Eur J Radiol 2013;82:e509-14. [Crossref] [PubMed]
- Crane CH, Koay EJ. Solutions that enable ablative radiotherapy for large liver tumors: Fractionated dose painting, simultaneous integrated protection, motion management, and computed tomography image guidance. Cancer 2016;122:1974-86. [Crossref] [PubMed]
- Memon K, Kulik L, Lewandowski RJ, et al. Radiographic response to locoregional therapy in hepatocellular carcinoma predicts patient survival times. Gastroenterology 2011;141:526-35, 535.e1-2.
- Shuster A, Huynh TJ, Rajan DK, et al. Response Evaluation Criteria in Solid Tumors (RECIST) criteria are superior to European Association for Study of the Liver (EASL) criteria at 1 month follow-up for predicting long-term survival in patients treated with transarterial chemoembolization before liver transplantation for hepatocellular cancer. J Vasc Interv Radiol 2013;24:805-12. [Crossref] [PubMed]
- Gillmore R, Stuart S, Kirkwood A, et al. EASL and mRECIST responses are independent prognostic factors for survival in hepatocellular cancer patients treated with transarterial embolization. J Hepatol 2011;55:1309-16. [Crossref] [PubMed]
- Hyder O, Marsh JW, Salem R, et al. Intra-arterial therapy for advanced intrahepatic cholangiocarcinoma: a multi-institutional analysis. Ann Surg Oncol 2013;20:3779-86. [Crossref] [PubMed]
- Camacho JC, Kokabi N, Xing M, et al. Modified response evaluation criteria in solid tumors and European Association for The Study of the Liver criteria using delayed-phase imaging at an early time point predict survival in patients with unresectable intrahepatic cholangiocarcinoma following yttrium-90 radioembolization. J Vasc Interv Radiol 2014;25:256-65. [Crossref] [PubMed]
- Bruix J, Sherman M, Llovet JM, et al. Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol 2001;35:421-30. [Crossref] [PubMed]
- Forner A, Ayuso C, Varela M, et al. Evaluation of tumor response after locoregional therapies in hepatocellular carcinoma: are response evaluation criteria in solid tumors reliable? Cancer 2009;115:616-23. [Crossref] [PubMed]
- Halappa VG, Bonekamp S, Corona-Villalobos CP, et al. Intrahepatic cholangiocarcinoma treated with local-regional therapy: quantitative volumetric apparent diffusion coefficient maps for assessment of tumor response. Radiology 2012;264:285-94. [Crossref] [PubMed]
- O'Connor JP, Aboagye EO, Adams JE, et al. Imaging biomarker roadmap for cancer studies. Nat Rev Clin Oncol 2017;14:169-86. [Crossref]
- Kim JH, Yoon HK, Ko GY, et al. Nonresectable combined hepatocellular carcinoma and cholangiocarcinoma: analysis of the response and prognostic factors after transcatheter arterial chemoembolization. Radiology 2010;255:270-7. [Crossref] [PubMed]
- Aliberti C, Benea G, Tilli M, et al. Chemoembolization (TACE) of unresectable intrahepatic cholangiocarcinoma with slow-release doxorubicin-eluting beads: preliminary results. Cardiovasc Intervent Radiol 2008;31:883-8. [Crossref] [PubMed]
- Herber S, Otto G, Schneider J, et al. Transarterial chemoembolization (TACE) for inoperable intrahepatic cholangiocarcinoma. Cardiovasc Intervent Radiol 2007;30:1156-65. [Crossref] [PubMed]
- Burger I, Hong K, Schulick R, et al. Transcatheter arterial chemoembolization in unresectable cholangiocarcinoma: initial experience in a single institution. J Vasc Interv Radiol 2005;16:353-61. [Crossref] [PubMed]
- Poggi G, Amatu A, Montagna B, et al. OEM-TACE: a new therapeutic approach in unresectable intrahepatic cholangiocarcinoma. Cardiovasc Intervent Radiol 2009;32:1187-92. [Crossref] [PubMed]
- Kiefer MV, Albert M, McNally M, et al. Chemoembolization of intrahepatic cholangiocarcinoma with cisplatinum, doxorubicin, mitomycin C, ethiodol, and polyvinyl alcohol: a 2-center study. Cancer 2011;117:1498-505. [Crossref] [PubMed]
- Vogl TJ, Naguib NN, Nour-Eldin NE, et al. Transarterial chemoembolization in the treatment of patients with unresectable cholangiocarcinoma: Results and prognostic factors governing treatment success. Int J Cancer 2012;131:733-40. [Crossref] [PubMed]
- Sieghart W, Hucke F, Peck-Radosavljevic M. Transarterial chemoembolization: modalities, indication, and patient selection. J Hepatol 2015;62:1187-95. [Crossref] [PubMed]
- Atiq OT, Kemeny N, Niedzwiecki D, et al. Treatment of unresectable primary liver cancer with intrahepatic fluorodeoxyuridine and mitomycin C through an implantable pump. Cancer 1992;69:920-4. [Crossref] [PubMed]
- Smith GW, Bukowski RM, Hewlett JS, et al. Hepatic artery infusion of 5-fluorouracil and mitomycin C in cholangiocarcinoma and gallbladder carcinoma. Cancer 1984;54:1513-6. [Crossref] [PubMed]
- Jarnagin WR, Schwartz LH, Gultekin DH, et al. Regional chemotherapy for unresectable primary liver cancer: results of a phase II clinical trial and assessment of DCE-MRI as a biomarker of survival. Ann Oncol 2009;20:1589-95. [Crossref] [PubMed]
- Kemeny NE, Schwartz L, Gonen M, et al. Treating primary liver cancer with hepatic arterial infusion of floxuridine and dexamethasone: does the addition of systemic bevacizumab improve results? Oncology 2011;80:153-9. [Crossref] [PubMed]
- Kim JH, Won HJ, Shin YM, et al. Radiofrequency ablation for recurrent intrahepatic cholangiocarcinoma after curative resection. Eur J Radiol 2011;80:e221-5. [Crossref] [PubMed]
- Yu MA, Liang P, Yu XL, et al. Sonography-guided percutaneous microwave ablation of intrahepatic primary cholangiocarcinoma. Eur J Radiol 2011;80:548-52. [Crossref] [PubMed]
- Xu HX, Wang Y, Lu MD, et al. Percutaneous ultrasound-guided thermal ablation for intrahepatic cholangiocarcinoma. Br J Radiol 2012;85:1078-84. [Crossref] [PubMed]
- Silk MT, Wimmer T, Lee KS, et al. Percutaneous ablation of peribiliary tumors with irreversible electroporation. J Vasc Interv Radiol 2014;25:112-8. [Crossref] [PubMed]
- Wahl DR, Stenmark MH, Tao Y, et al. Outcomes After Stereotactic Body Radiotherapy or Radiofrequency Ablation for Hepatocellular Carcinoma. J Clin Oncol 2016;34:452-9. [Crossref] [PubMed]
- Mouli S, Memon K, Baker T, et al. Yttrium-90 radioembolization for intrahepatic cholangiocarcinoma: safety, response, and survival analysis. J Vasc Interv Radiol 2013;24:1227-34. [Crossref] [PubMed]
- Kim YC, Kim YH, Um SH, et al. Usefulness of Bremsstrahlung Images after Intra-arterial Y-90 Resin Microphere Radioembolization for Hepatic Tumors. Nucl Med Mol Imaging 2011;45:59-67. [Crossref] [PubMed]
- Mikell JK, Mahvash A, Siman W, et al. Selective Internal Radiation Therapy With Yttrium-90 Glass Microspheres: Biases and Uncertainties in Absorbed Dose Calculations Between Clinical Dosimetry Models. Int J Radiat Oncol Biol Phys 2016;96:888-96. [Crossref] [PubMed]
- Strigari L, Sciuto R, Rea S, et al. Efficacy and toxicity related to treatment of hepatocellular carcinoma with 90Y-SIR spheres: radiobiologic considerations. J Nucl Med 2010;51:1377-85. [Crossref] [PubMed]
- Vouche M, Habib A, Ward TJ, et al. Unresectable solitary hepatocellular carcinoma not amenable to radiofrequency ablation: multicenter radiology-pathology correlation and survival of radiation segmentectomy. Hepatology 2014;60:192-201. [Crossref] [PubMed]
- Fukumitsu N, Sugahara S, Nakayama H, et al. A prospective study of hypofractionated proton beam therapy for patients with hepatocellular carcinoma. Int J Radiat Oncol Biol Phys 2009;74:831-6. [Crossref] [PubMed]
- Kawashima M, Furuse J, Nishio T, et al. Phase II study of radiotherapy employing proton beam for hepatocellular carcinoma. J Clin Oncol 2005;23:1839-46. [Crossref] [PubMed]
- Mizumoto M, Okumura T, Hashimoto T, et al. Proton beam therapy for hepatocellular carcinoma: a comparison of three treatment protocols. Int J Radiat Oncol Biol Phys 2011;81:1039-45. [Crossref] [PubMed]
- Nakajima T, Kondo Y, Miyazaki M, et al. A histopathologic study of 102 cases of intrahepatic cholangiocarcinoma: histologic classification and modes of spreading. Hum Pathol 1988;19:1228-34. [Crossref] [PubMed]
- Lim JH. Cholangiocarcinoma: morphologic classification according to growth pattern and imaging findings. AJR Am J Roentgenol 2003;181:819-27. [Crossref] [PubMed]
- Bhuiya MR, Nimura Y, Kamiya J, et al. Clinicopathologic studies on perineural invasion of bile duct carcinoma. Annals of Surgery 1992;215:344-9. [Crossref] [PubMed]
- Soyer P, Bluemke DA, Reichle R, et al. Imaging of intrahepatic cholangiocarcinoma: 1. Peripheral cholangiocarcinoma. AJR Am J Roentgenol 1995;165:1427-31. [Crossref] [PubMed]
- Chung YE, Kim MJ, Park YN, et al. Varying Appearances of Cholangiocarcinoma: Radiologic-Pathologic Correlation. RadioGraphics 2009;29:683-700. [Crossref] [PubMed]
- Maetani Y, Itoh K, Watanabe C, et al. MR imaging of intrahepatic cholangiocarcinoma with pathologic correlation. AJR Am J Roentgenol 2001;176:1499-507. [Crossref] [PubMed]
- Sainani NI, Catalano OA, Holalkere NS, et al. Cholangiocarcinoma: current and novel imaging techniques. Radiographics 2008;28:1263-87. [Crossref] [PubMed]
- Tacher V, Lin M, Duran R, et al. Comparison of Existing Response Criteria in Patients with Hepatocellular Carcinoma Treated with Transarterial Chemoembolization Using a 3D Quantitative Approach. Radiology 2016;278:275-84. [Crossref] [PubMed]
- Chapiro J, Duran R, Lin M, et al. Early survival prediction after intra-arterial therapies: a 3D quantitative MRI assessment of tumour response after TACE or radioembolization of coloreFctal cancer metastases to the liver. Eur Radiol 2015;25:1993-2003. [Crossref] [PubMed]
- Andersen JB, Spee B, Blechacz BR, et al. Genomic and genetic characterization of cholangiocarcinoma identifies therapeutic targets for tyrosine kinase inhibitors. Gastroenterology 2012;142:1021-31.e15. [Crossref] [PubMed]
- Jain A, Javle M. Molecular profiling of biliary tract cancer: a target rich disease. J Gastrointest Oncol 2016;7:797-803. [Crossref] [PubMed]